Thymosin Alpha-1
$395.00
The Klow Blend combines four research peptides, BPC-157, TB-500, KPV, and GHK-Cu, formulated for studies on cellular repair, immune modulation, and extracellular matrix regulation. BPC-157 is a 15 amino acid gastric peptide fragment that modulates nitric oxide and growth factor pathways. TB-500, a segment of Thymosin Beta-4, regulates actin polymerization and tissue remodeling. KPV, derived from the C-terminal region of alpha-melanocyte-stimulating hormone, modulates NF-κB and cytokine signaling. GHK-Cu, a copper(II)-complexed tripeptide, influences metalloprotein activity and redox balance. Together, these peptides are used in research examining regeneration, inflammation, and peptide–metal interaction mechanisms.
For research use only. Not for human consumption.
References:
Sikiric P et al., Curr Pharm Des, 2018 24(18):2002–2030
Goldstein AL et al., Ann NY Acad Sci, 2007 1112:148–154
Getting SJ et al., J Immunol, 1999 163(10):5959–5967
Pickart L et al., Biochim Biophys Acta, 2015 1850(8):1863–1885
Overview
Thymosin Alpha-1 (Tα1; also referenced as thymalfasin in portions of the literature) is a 28-amino-acid peptide originally characterized from thymic peptide fractions and widely used as a laboratory reagent in experimental immunology and cell signaling research. Published studies include in vitro cellular systems and in vivo animal models that evaluate Tα1-driven modulation of pattern-recognition receptor signaling, antigen-presenting cell maturation, cytokine network behavior, and downstream transcriptional programs.
Within research settings, Tα1 is commonly studied for its effects on innate/adaptive signaling crosstalk, dendritic cell functional state, T-cell polarization dynamics, and pathway-level biomarkers measured under controlled experimental conditions.
Biochemical Characteristics
 Source: PubChem
Source: PubChem
Sequence: Ser-Asp-Ala-Ala-Val-Asp-Thr-Ser-Ser-Glu-Ile-Thr-Thr-Lys-Asp-Leu-Lys-Glu-Lys-Lys-Glu-Val-Val-Glu-Glu-Ala-Glu-Asn
Molecular Formula: C129H215N33O55
Molecular Weight: 3108.315 g/mol
PubChem CID: 16130571
CAS Number: 62304-98-7
Synonyms: Thymalfasin
Research Applications
- Innate immune signaling: experimental systems evaluating Toll-like receptor (TLR)-associated signaling, interferon-linked transcriptional responses, and cytokine network dynamics under defined stimulation conditions.
- Antigen-presenting cell biology: dendritic cell maturation, antigen processing/presentation markers, and downstream effects on T-cell priming in cell culture and animal models.
- T-cell polarization and adaptive signaling: assays measuring Th1/Th2-associated cytokine patterns, CD4/CD8 compartment behavior, and activation marker profiles in controlled experimental paradigms.
- Host–pathogen model endpoints: in vivo and in vitro models that quantify immune pathway biomarkers during bacterial, viral, or fungal challenge studies.
- Neuroimmune interactions: preclinical models evaluating immune-linked gene programs associated with neurodevelopmental and neuroinflammatory signaling readouts.
- Oncology research endpoints: cell-based proliferation/migration assays and animal tumor-model readouts used to explore immune-modulated pathway behavior in experimental settings.
- Enzymology and peptide–protein interaction studies: biochemical screens assessing ACE-inhibitory activity and redox-associated markers as measured endpoints in defined assay conditions.
Pathway / Mechanistic Context
Mechanistic literature describes Thymosin Alpha-1 as a peptide capable of shifting immune signaling states through coordinated effects on antigen-presenting cells and downstream T-cell response programs. In dendritic cell systems, Tα1 has been reported to engage Toll-like receptor-linked signaling pathways and to promote functional maturation phenotypes, with downstream effects on Th1-associated cytokine profiles measured in experimental models [5] [6].
In immune challenge paradigms, reported endpoints include cytokine production patterns, interferon-linked transcriptional markers, activation-state signatures, and tolerance-associated readouts under defined stimulation conditions [1]. In vaccine-adjuvant research models, studies evaluate antigen-specific response markers using inactivated antigen platforms and quantify immunogenicity-related laboratory endpoints over defined time courses [2].
Neuroimmune research also evaluates Tα1-associated shifts in systemic immune bias and corresponding transcriptional programs linked to neurogenesis and synaptic pathway markers in developing mouse models, using gene-expression profiling and behavioral assay outputs as experimental readouts [4].
Additional biochemical studies examine Tα1 in enzyme-target assays, including angiotensin-converting enzyme (ACE) inhibitory activity and redox-associated biomarkers (e.g., ROS-linked and antioxidant enzyme activity measures) in controlled laboratory systems [10] [11].
Preclinical Research Summary
1. Immune Signaling Modulation in Experimental Models
Reviews and primary studies describe Tα1-associated changes in immune signaling states, including cytokine profile shifts and activation-marker endpoints in controlled experimental systems [1]. In vaccine-adjuvant models, recombinant fusion peptide constructs incorporating Tα1 have been evaluated for immune-adjuvant properties using inactivated antigen preparations, with study endpoints including antigen-specific response markers quantified in preclinical settings [2].
2. Dendritic Cell Maturation and Antifungal Response Models
In experimental antifungal models, dendritic cell maturation and Toll-like receptor signaling are reported as mechanistic nodes influenced by Tα1, with downstream Th1-associated resistance markers measured as outcomes in defined study designs [5]. Additional work discusses Tα1 as an endogenous regulator of inflammation, immunity, and tolerance, focusing on pathway-level signals and immune-network behavior as experimental endpoints [6].
3. Neuroimmune Gene Programs in Developing Mouse Models
Preclinical mouse studies report that systemic immune-bias modulation is associated with measured changes in neurogenesis-linked readouts and cognition-associated assay outcomes in developing animals, with gene-expression analyses used to define affected pathways [4].
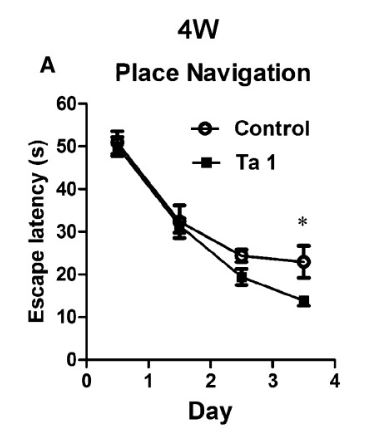 A. 4-week-old mice given thymosin alpha-1 learn how to escape from mazes faster.
A. 4-week-old mice given thymosin alpha-1 learn how to escape from mazes faster.
Source: PubMed
4. Cell-Based Oncology Endpoints and Immune-Linked Biomarkers
Cell culture studies in epithelial adenocarcinoma model systems report Tα1-associated changes in proliferation and migration assays, alongside redox-related laboratory endpoints [11]. Separate preclinical investigations describe long-acting Tα1 fusion constructs evaluated in vivo, with measured outcomes including tumor-model endpoints and immune cell marker changes in controlled study designs [14] [15].
5. Enzyme-Target Assays and Redox-Associated Readouts
Biochemical studies report ACE-inhibitory activity for Tα1 in defined assay conditions, alongside antioxidant/ROS-associated endpoints measured in laboratory systems [10].
6. Additional Preclinical Contexts in the Cited Literature
The referenced citations include a broad range of experimental contexts (e.g., infection-model research, inflammation/tolerance frameworks, and biomarker studies). Where present in the literature, these investigations are characterized by preclinical endpoints, pathway-level biomarkers, and controlled experimental designs suitable for mechanistic interpretation [8] [9] [16] [17].
Form & Analytical Testing
Thymosin Alpha-1 is a 28-residue peptide (Tα1) supplied as a research material intended for controlled laboratory workflows. Laboratories may reference sequence and registry identifiers (CAS, PubChem CID) for internal documentation and materials qualification.
For experimental planning and quality workflows, peptide identity and analytical attributes are commonly verified using characterization approaches appropriate to peptide materials, aligned to internal methods and study requirements.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Allan L. Goldstein, MD, Allan L. Goldstein is professor and Catharine B. & William McCormick Chair of the department of Biochemistry and Molecular Biology at The George Washington University School of Medicine and Health Sciences, where he has served since 1978. Thymosins were discovered in the mid 1960’s, when Allan Goldstein from the Laboratory of Abraham White at the Albert Einstein College of Medicine in New York studied the role of the thymus in development of the vertebrate immune system. He is a world-renowned authority on the thymus gland and the workings of the immune system, and co-discoverer of the thymosins. Dr. Goldstein is the author of over 400 scientific articles in professional journals, the inventor on more than 15 U.S. Patents, and the editor of several books in the fields of biochemistry, biomedicine, immunology and neuro-science. He is on the editorial boards of numerous scientific and medical journals and has been a consultant to many re-search organizations in industry and government; co-founder of The Institute for Advanced Studies in Aging and Geriatric Medicine, a non-profit research and educational institute; a member of the Board of Trustees of the Albert Sabin Vaccine Institute; and serves as the Chairman of the Board of RegeneRx Biopharmaceuticals. Dr. Goldstein received his B.S. from Wagner College in 1959 and his M.S. and Ph.D. from Rutgers University in 1964. He served as a faculty member of the Albert Einstein College of Medicine from 1964 to 1972, and moved to the University of Texas Medical Branch in Galveston in 1972 as professor and director of the division of Biochemistry.
Allan L. Goldstein, MD is being referenced as one of the leading scientists involved in the research and development of Thymosin Alpha 1 and other Thymosins. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Goldstein is listed in [7] under the referenced citations.
Referenced Citations
- R. King and C. Tuthill, “Immune Modulation with Thymosin Alpha 1 Treatment,” Vitam. Horm., vol. 102, pp. 151–178, 2016.
- C. Zhang, J. Zhou, K. Cai, W. Zhang, C. Liao, and C. Wang, “Gene cloning, expression and immune adjuvant properties of the recombinant fusion peptide Tα1-BLP on avian influenza inactivate virus vaccine,” Microb. Pathog., vol. 120, pp. 147–154, Jul. 2018.
- F. Pei, X. Guan, and J. Wu, “Thymosin alpha 1 treatment for patients with sepsis,” Expert Opin. Biol. Ther., vol. 18, no. sup1, pp. 71–76, 2018.
- G. Wang et al., “Immunopotentiator Thymosin Alpha-1 Promotes Neurogenesis and Cognition in the Developing Mouse via a Systemic Th1 Bias,” Neurosci. Bull., vol. 33, no. 6, pp. 675–684, Dec. 2017.
- L. Romani et al., “Thymosin α 1 activates dendritic cells for antifungal Th1 resistance through Toll-like receptor signaling,” Blood, vol. 103, no. 11, pp. 4232–4239, Jun. 2004.
- L. Romani et al., “Thymosin alpha1: an endogenous regulator of inflammation, immunity, and tolerance,” Ann. N. Y. Acad. Sci., vol. 1112, pp. 326–338, Sep. 2007.
- A. L. Goldstein and A. L. Goldstein, “From lab to bedside: emerging clinical applications of thymosin alpha 1,” Expert Opin. Biol. Ther., vol. 9, no. 5, pp. 593–608, May 2009.
- C. Matteucci et al., “Thymosin alpha 1 and HIV-1: recent advances and future perspectives,” Future Microbiol., vol. 12, pp. 141–155, 2017.
- C. Matteucci et al., “Thymosin α 1 potentiates the release by CD8(+) cells of soluble factors able to inhibit HIV-1 and human T lymphotropic virus 1 infection in vitro,” Expert Opin. Biol. Ther., vol. 15 Suppl 1, pp. S83-100, 2015.
- J. Kharazmi-Khorassani, A. Asoodeh, and H. Tanzadehpanah, “Antioxidant and angiotensin-converting enzyme (ACE) inhibitory activity of thymosin alpha-1 (Thα1) peptide,” Bioorganic Chem., vol. 87, pp. 743–752, Jun. 2019.
- J. Kharazmi-Khorassani and A. Asoodeh, “Thymosin alpha-1; a natural peptide inhibits cellular proliferation, cell migration, the level of reactive oxygen species and promotes the activity of antioxidant enzymes in human lung epithelial adenocarcinoma cell line (A549),” Environ. Toxicol., May 2019.
- M. Maio et al., “Large randomized study of thymosin alpha 1, interferon alfa, or both in combination with dacarbazine in patients with metastatic melanoma,” J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol., vol. 28, no. 10, pp. 1780–1787, Apr. 2010.
- R. Danielli, E. Fonsatti, L. Calabrò, A. M. Di Giacomo, and M. Maio, “Thymosin α1 in melanoma: from the clinical trial setting to the daily practice and beyond,” Ann. N. Y. Acad. Sci., vol. 1270, pp. 8–12, Oct. 2012.
- X. Shen et al., “Generation of a novel long-acting thymosin alpha1-Fc fusion protein and its efficacy for the inhibition of breast cancer in vivo,” Biomed. Pharmacother. Biomedecine Pharmacother., vol. 108, pp. 610–617, Dec. 2018.
- F. Wang, T. Yu, H. Zheng, and X. Lao, “Thymosin Alpha1-Fc Modulates the Immune System and Down-regulates the Progression of Melanoma and Breast Cancer with a Prolonged Half-life,” Sci. Rep., vol. 8, no. 1, p. 12351, Aug. 2018.
- Y. Xu et al., “Thymosin Alpha-1 Inhibits Complete Freund’s Adjuvant-Induced Pain and Production of Microglia-Mediated Pro-inflammatory Cytokines in Spinal Cord,” Neurosci. Bull., Feb. 2019.
- L. Romani et al., “Thymosin α1 represents a potential potent single-molecule-based therapy for cystic fibrosis,” Nat. Med., vol. 23, no. 5, pp. 590–600, May 2017.
- P. F. Day, M. Duggal, and H. Nazzal, “Interventions for treating traumatised permanent front teeth: avulsed (knocked out) and replanted,” Cochrane Database Syst. Rev., vol. 2, p. CD006542, 05 2019.
- M. Schmidt et al., “Design of a substrate-tailored peptiligase variant for the efficient synthesis of thymosin-α1,” Org. Biomol. Chem., vol. 16, no. 4, pp. 609–618, 24 2018.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat, or cure any disease or condition. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.






