Tesamorelin 2mg (10 Vial Kit)
$110.00
Tesamorelin is a synthetic research peptide and stabilized analog of growth hormone–releasing hormone (GHRH) designed to selectively activate the GHRH receptor and stimulate endogenous growth hormone secretion in experimental models. Preclinical and clinical research has focused on its role in modulating somatotropic signaling, lipid metabolism, and visceral adipose regulation through cAMP-dependent pituitary pathways. Tesamorelin is utilized in research investigating hypothalamic–pituitary axis dynamics, metabolic signaling, neuroendocrine regulation, and growth hormone–mediated tissue effects.
For research use only. Not for human consumption.
References:
Falutz J et al., N Engl J Med, 2010 363(6):528–539
Stanley TL et al., J Clin Endocrinol Metab, 2011 96(6):E1022–E1032
Grinspoon SK et al., J Clin Endocrinol Metab, 2014 99(11):4099–4107
Overview
Tesamorelin is a synthetic growth hormone–releasing hormone (GHRH) analogue used as a laboratory tool compound to investigate growth hormone–releasing hormone receptor (GHRHR) signaling and downstream endocrine pathway dynamics in preclinical experimental systems. The molecule incorporates an additional trans-3-hexanoic acid group, a peptide-chemistry modification often used to modulate physicochemical stability and proteolytic susceptibility in biological matrices.
In RUO settings, tesamorelin is applied in mechanistic studies of receptor-coupled signaling, endocrine feedback circuitry, and GH/IGF-axis–associated molecular readouts in controlled in-vitro assays and in-vivo animal models.
Biochemical Characteristics
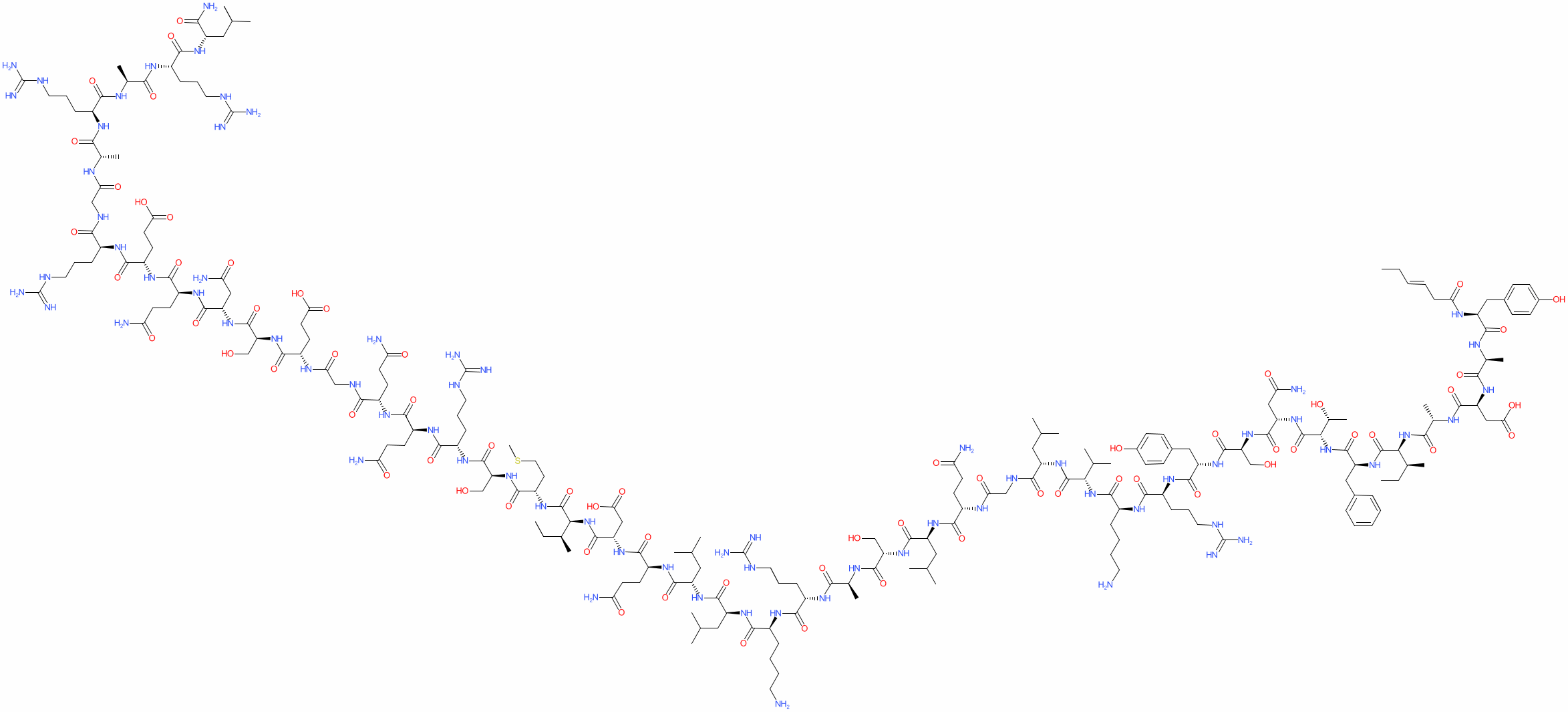 Sequence (as provided): Unk-Tyr-Ala-Asp-Ala-Ile-Phe-Thr-Asn-Ser-Tyr-Arg-Lys-Val-Leu-Gly-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Met-Ser-Arg-Gln-Gln-Gly-Glu-Ser-Asn-Gln-Glu-Arg-Gly-Ala-Arg-Ala-Arg-Leu
Sequence (as provided): Unk-Tyr-Ala-Asp-Ala-Ile-Phe-Thr-Asn-Ser-Tyr-Arg-Lys-Val-Leu-Gly-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Met-Ser-Arg-Gln-Gln-Gly-Glu-Ser-Asn-Gln-Glu-Arg-Gly-Ala-Arg-Ala-Arg-LeuMolecular Formula: C223H370N72O69S
Molecular Weight: 5195.908 g/mol
PubChem CID: 44147413
CAS Number: 901758-09-6
Tesamorelin is engineered as a GHRH-family peptide analogue to support reproducible handling and exposure profiles in experimental workflows. In laboratory studies, such modifications may be leveraged to evaluate stability-sensitive endpoints, ligand–receptor interaction consistency, and dose–response behavior within defined assay conditions.
Research Applications
As a GHRH analogue, tesamorelin is used in laboratory research to probe GHRHR activation and downstream signaling associated with GH/IGF-axis regulation. In experimental contexts, GHRHR engagement is commonly associated with GPCR-mediated signaling (often Gs/cAMP-linked), modulation of kinase activity, and transcriptional programs that influence endocrine signaling dynamics.
Common RUO applications (non-exhaustive) include:
- GHRHR pharmacology: ligand binding, potency, and receptor-activation assays
- Second-messenger studies: adenylate cyclase/cAMP-linked signaling readouts and downstream transcriptional responses
- GH/IGF-axis pathway mapping in controlled in-vitro systems and in-vivo animal models
- Endocrine feedback and signaling kinetics experiments (including receptor desensitization concepts)
- Exploratory biomarker workflows in metabolic and neuroendocrine research models
The trans-3-hexanoic acid modification is used in peptide engineering to support consistent experimental performance and facilitate mechanistic interrogation of receptor signaling and feedback regulation under controlled laboratory conditions.
Pathway / Mechanistic Context
Tesamorelin is studied as a GHRH-pathway probe that signals through the growth hormone–releasing hormone receptor (GHRHR), a class B G protein–coupled receptor. In canonical models, receptor engagement is associated with adenylate cyclase activation and increased intracellular cAMP, supporting downstream phosphorylation events and regulated gene-expression programs relevant to somatotroph and endocrine-axis function.
In research systems, downstream signaling may be evaluated via endocrine readouts, transcriptional signatures, kinase-node cross-talk, and biomarker relationships tied to GH/IGF-axis regulation and metabolic or neuroendocrine endpoints.
Preclinical Research Summary
1. Experimental Modeling of Visceral Adiposity & Lipid Biomarkers
Published studies have evaluated tesamorelin and related growth hormone–releasing factor analogues in research cohorts using metabolic endpoints and biomarker readouts. Reported analyses include associations between changes in visceral adipose tissue measures and lipid parameters such as triglycerides [3], [4]. These citations are provided strictly for scientific background and do not imply any intended medical, diagnostic, or therapeutic use.
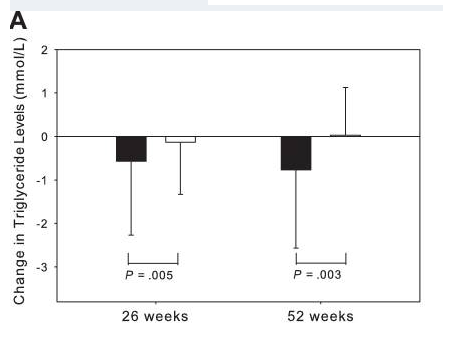 Figure included as an educational reference to the cited publication record.
Figure included as an educational reference to the cited publication record.
Source: PubMed
2. Endocrine Axis Research in Chronic Infection Models
Reviews and mechanistic discussions have described endocrine and metabolic alterations observed in chronic infection research settings, including reported changes in growth hormone axis parameters [5]. In RUO laboratories, tesamorelin may be used as a receptor-directed probe to evaluate signaling relationships and endocrine feedback behavior under controlled experimental conditions.
3. Peripheral Nerve Biology & Growth Hormone Axis Augmentation (Preclinical Context)
The growth hormone axis has been discussed in preclinical literature as a modulator of cellular environments relevant to peripheral nerve injury models and regenerative biology research. Reviews have summarized potential mechanistic links between endocrine signaling and experimental outcomes in nerve repair paradigms [6]. These discussions support hypothesis generation for mechanistic studies (e.g., growth factor signaling, Schwann cell-associated pathways, and axonal outgrowth processes) in non-clinical settings.
4. Neuroendocrine Signaling & Biomarker Readouts in Cognitive Research
GHRH analogues have been evaluated in published research using biomarker and neurochemical readouts, including spectroscopy-associated measures and neurotransmitter-related endpoints, as part of broader investigations into neuroendocrine signaling in aging models [7]. References are provided for pathway context only.
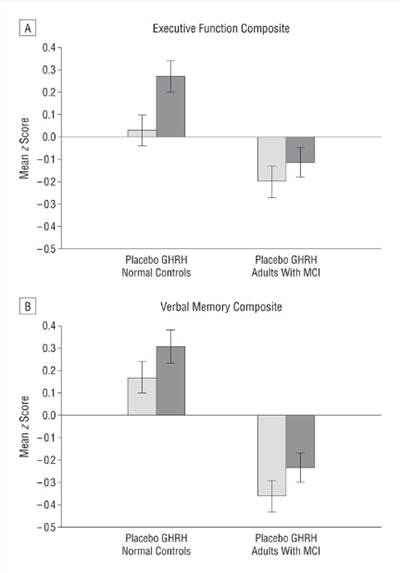 Figure included as an educational reference to the cited publication record.
Figure included as an educational reference to the cited publication record.
Source: PubMed
All research summaries above reflect preclinical, translational, and laboratory observations and are provided solely to support experimental planning and mechanistic discussion.
Form & Analytical Testing
Tesamorelin supplied for research is commonly prepared as a synthetic peptide suitable for laboratory handling. Product identity and purity are typically verified using established analytical techniques such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS). Batch-specific documentation may be provided to support research reproducibility and quality review.
Materials are intended to support controlled experimental workflows and downstream characterization using standard laboratory assays.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Referenced Citations
- Clinical Review Report: Tesamorelin (Egrifta). Ottawa (ON): Canadian Agency for Drugs and Technologies in Health, 2016.
- A. Mangili, J. Falutz, J.-C. Mamputu, M. Stepanians, and B. Hayward, “Predictors of Treatment Response to Tesamorelin, a Growth Hormone-Releasing Factor Analog, in HIV-Infected Patients with Excess Abdominal Fat,” PloS One, vol. 10, no. 10, p. e0140358, 2015. [PubMed]
- J. Falutz et al., “Metabolic effects of a growth hormone-releasing factor in patients with HIV,” N. Engl. J. Med., vol. 357, no. 23, pp. 2359–2370, Dec. 2007. [NEJM]
- T. L. Stanley et al., “Reduction in visceral adiposity is associated with an improved metabolic profile in HIV-infected patients receiving tesamorelin,” Clin. Infect. Dis., vol. 54, no. 11, pp. 1642–1651, Jun. 2012. [PubMed]
- V. Rochira and G. Guaraldi, “Growth hormone deficiency and human immunodeficiency virus,” Best Pract. Res. Clin. Endocrinol. Metab., vol. 31, no. 1, pp. 91–111, 2017. [PubMed]
- S. H. Tuffaha et al., “Therapeutic augmentation of the growth hormone axis to improve outcomes following peripheral nerve injury,” Expert Opin. Ther. Targets, vol. 20, no. 10, pp. 1259–1265, Oct. 2016. [PubMed]
- S. D. Friedman et al., “Growth hormone-releasing hormone effects on brain γ-aminobutyric acid levels in mild cognitive impairment and healthy aging,” JAMA Neurol., vol. 70, no. 7, pp. 883–890, Jul. 2013. [PubMed]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.