P21 (P021) 5mg
$120.00
P21 is a synthetic research peptide derived from a β-amyloid sequence fragment (Aβ₁₆–₂₀), designed to modulate tau phosphorylation and neuroprotective signaling in preclinical models. Studies demonstrate its potential to influence synaptic plasticity and oxidative stress resistance through regulation of GSK-3β and MAPK pathways. P21 is commonly utilized to explore neuroregenerative and neuroprotective mechanisms in peptide-mediated cellular research.
For research use only. Not for human consumption.
References:
Seeger TF et al., Neurobiol Aging, 1997 18(5):619–630
Smith DL et al., J Neurosci Res, 2009 87(2):274–284
Puzzo D et al., Nat Commun, 2017 8:14727
Overview
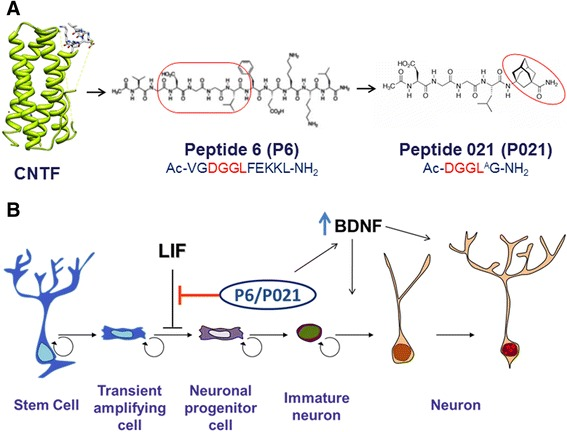
P21 (also referred to as P021 in the scientific literature) is a synthetic, sequence-defined peptide derived from a short epitope within ciliary neurotrophic factor (CNTF). CNTF is an endogenous cytokine-like protein that has been extensively investigated in preclinical neuroscience as a regulator of neuronal differentiation, neurogenic signaling, and transcriptional balance in experimental systems. P21 was designed as a reduced-length molecular probe to facilitate mechanistic investigation of CNTF-associated pathways in vitro and in vivo animal models.
Unlike full-length CNTF, which presents experimental limitations related to molecular size, stability, and immunogenicity, P21 represents a truncated, chemically modified construct optimized for laboratory research applications. P21 is distinct from cerebrolysin and should not be considered chemically or functionally equivalent; rather, it is a defined synthetic peptide intended for pathway-level and molecular studies.
Development of P21 was motivated by the need for a stable, minimal peptide tool capable of interrogating CNTF-linked signaling networks without reliance on recombinant protein delivery. All descriptions herein reflect preclinical research contexts only.
Biochemical Characteristics
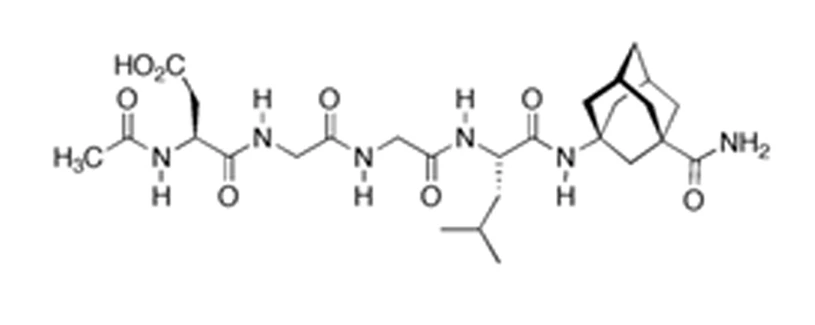
Sequence: DGGL–adamantane–G
Molecular Formula: C30H54N6O5
Molecular Weight: 578.3 g/mol
Synonyms: P021, Peptide 021
Source: Nature
P21 consists of a tetrapeptide core derived from CNTF amino acid residues 148–151 with a covalently linked adamantane moiety. Adamantylation is a commonly employed modification in peptide chemistry to enhance resistance to proteolytic degradation and to support experimental evaluation of central nervous system–associated signaling pathways in animal models. The defined molecular structure of P21 enables its use as a reproducible probe in peptide–pathway interaction studies.
Research Applications
How was P21 Developed?
P21 was developed using epitope-mapping strategies designed to identify minimal peptide sequences capable of modulating CNTF-associated signaling environments. Antibodies directed against CNTF receptor–associated epitopes were used to define target interaction regions and to screen synthetic peptide candidates for interference with antibody binding under controlled experimental conditions. This process led to identification of DGGL-based peptide motifs suitable for mechanistic studies.
Subsequent chemical modification through addition of an adamantylated glycine residue was employed to enhance experimental stability and persistence in preclinical model systems. These features render P21 suitable for controlled laboratory investigation of neurotrophic signaling pathways.
Pathway / Mechanistic Context
Preclinical investigations in rodent models have associated P21 exposure with altered cellular proliferation and differentiation markers within the dentate gyrus of the hippocampal formation. The dentate gyrus is a well-characterized neurogenic niche frequently used to study transcriptional and signaling events associated with neuronal lineage progression in adult mammals.
Experimental observations include increased incorporation of bromodeoxyuridine (BrdU) and elevated expression of neuronal markers such as NeuN in defined hippocampal subregions. These readouts are interpreted as indicators of altered cell-cycle dynamics and neuronal maturation under experimental conditions.
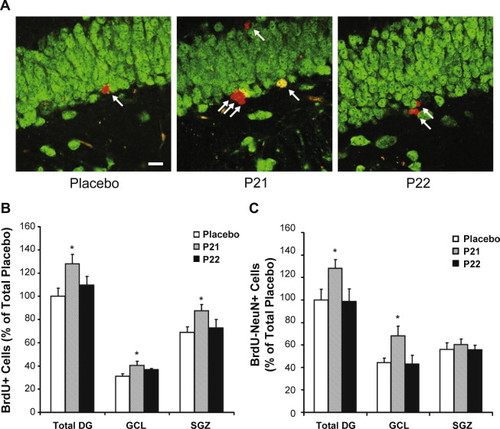 Representative BrdU and NeuN immunolabeling patterns reported in rodent experimental models.
Representative BrdU and NeuN immunolabeling patterns reported in rodent experimental models.
Signaling Network Associations
Mechanistic studies describe modulation of signaling pathways involving leukemia inhibitory factor (LIF), JAK/STAT-associated transcriptional regulation, and neurotrophin-linked signaling cascades. Inhibition of LIF-associated signaling has been discussed as a permissive factor for differentiation-associated gene expression in neural precursor populations.
 Additional experimental reports describe altered expression or activity of molecular intermediates associated with BDNF, PI3K, and GSK-3β pathways. These nodes are widely studied in neuroscience research as regulators of cytoskeletal organization, synaptic architecture, and transcriptional plasticity.
Additional experimental reports describe altered expression or activity of molecular intermediates associated with BDNF, PI3K, and GSK-3β pathways. These nodes are widely studied in neuroscience research as regulators of cytoskeletal organization, synaptic architecture, and transcriptional plasticity.
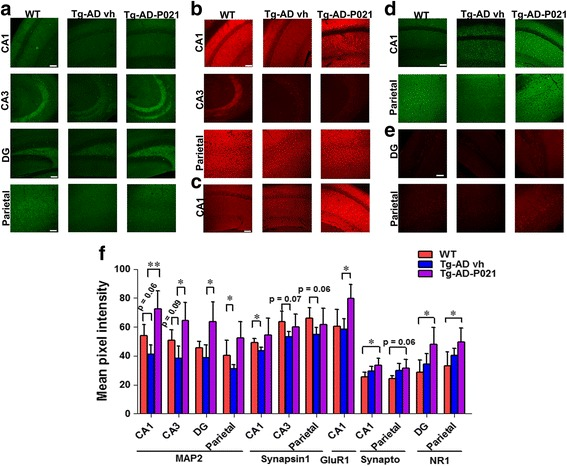
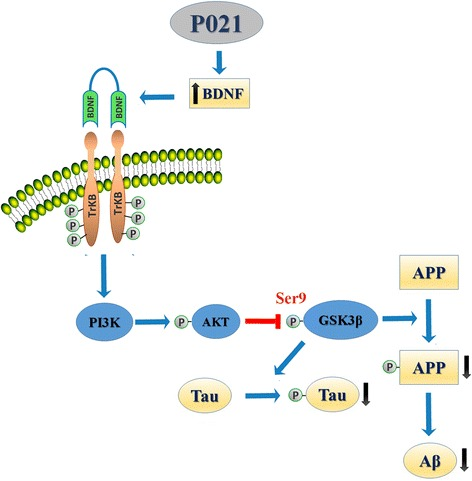 Changes in expression of synaptic markers including MAP2, synapsin I, GluR1, and NR1 have been reported in animal models exposed to P21. These proteins are commonly employed as molecular readouts in studies of synaptogenesis and neuronal connectivity in both baseline and disease-model contexts.
Changes in expression of synaptic markers including MAP2, synapsin I, GluR1, and NR1 have been reported in animal models exposed to P21. These proteins are commonly employed as molecular readouts in studies of synaptogenesis and neuronal connectivity in both baseline and disease-model contexts.
Preclinical Research Summary
P21 is a synthetic CNTF-derived peptide used exclusively as a laboratory research tool for investigating neurotrophic signaling, neurogenesis-associated transcriptional programs, and pathway-level regulation in preclinical experimental systems. All reported findings derive from in vitro assays or in vivo animal studies and are presented for scientific and educational purposes only.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Khalid Iqbal is Professor and Chairman, Department of Neurochemistry, at the New York State Institute for Basic Research in Developmental Disabilities, Staten Island, New York. He received his Ph.D. in Biochemistry in 1969 from the University of Edinburgh, Edinburgh, U.K. Dr. Iqbal was the first to describe in 1974 the bulk isolation and protein composition of neurofibrillary tangles/paired helical filaments (PHF) from Alzheimer disease brains. In 1986 he, along with Dr. Inge Grundke-Iqbal, discovered that the PHF protein and the microtubule-associated protein tau are the same and that tau in PHF is hyperphosphorylated. Their search for an upstream-to-tau-pathology event led them to neurotrophic factors. In 1999 they discovered that CNTF could neutralize the FGF-2-mediated tau hyperphosphorylation in adult rat hippocampal neuroprogenitor cells and then, in 2003, they demonstrated that the pharmacologic enhancement of the dentate gyrus neurogenesis could improve the cognitive performance in adult rats. These pioneering studies led Drs. Iqbal and Grundke-Iqbal to the development of CNTF peptidergic compounds and a novel therapeutic approach that involved shifting the balance from neurodegeneration to the regeneration of the brain. They have shown that the CNTF peptidergic compounds can rescue cognitive impairment by rescuing the neurogenesis and the neuronal plasticity deficits in rodent models of familial and sporadic Alzheimer disease and Down syndrome.
Dr. Iqbal is the recipient of many prestigious honors and awards, including the Potamkin Prize for Alzheimer Disease research from the American Academy of Neurology, and the Zenith Award from the Alzheimer’s Association, U.S.A. He founded and chaired the biennial International Conference on Alzheimer’s Disease from 1988 to 2008. In 2007, the Alzheimer’s Association, U.S.A. established a Khalid Iqbal Life Time Achievement Award for Alzheimer’s Disease Research, which is given out annually at the International Conference on Alzheimer’s Disease (ICAD) to a senior established Alzheimer disease researcher. Dr. Iqbal has authored over 300 scientific papers in prestigious American and international scientific journals and edited seven books on research advances in Alzheimer disease and related neurodegenerative disorders. He currently serves on the editorial boards of several journals.
Khalid Iqbal, Ph.D. is being referenced as one of the leading scientists involved in the research and development of P-21. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Igbal is listed in [5] under the referenced citations.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.