BPC-157, TB-500, KPV, GHK-Cu 80mg (Klow Blend) FREE Shipping New
$165.00
The Klow Blend combines four research peptides, BPC-157, TB-500, KPV, and GHK-Cu, formulated for studies on cellular repair, immune modulation, and extracellular matrix regulation. BPC-157 is a 15 amino acid gastric peptide fragment that modulates nitric oxide and growth factor pathways. TB-500, a segment of Thymosin Beta-4, regulates actin polymerization and tissue remodeling. KPV, derived from the C-terminal region of alpha-melanocyte-stimulating hormone, modulates NF-κB and cytokine signaling. GHK-Cu, a copper(II)-complexed tripeptide, influences metalloprotein activity and redox balance. Together, these peptides are used in research examining regeneration, inflammation, and peptide–metal interaction mechanisms.
For research use only. Not for human consumption.
References:
Sikiric P et al., Curr Pharm Des, 2018 24(18):2002–2030
Goldstein AL et al., Ann NY Acad Sci, 2007 1112:148–154
Getting SJ et al., J Immunol, 1999 163(10):5959–5967
Pickart L et al., Biochim Biophys Acta, 2015 1850(8):1863–1885
Overview
The BPC-157 + TB-500 (Thymosin Beta-4) + KPV + GHK-Cu Blend is a multi-peptide research formulation designed to support investigation into synergistic mechanisms of tissue repair, inflammation regulation, and regenerative biology. Each peptide included in this blend has independently demonstrated robust activity in preclinical models of wound healing, immune modulation, angiogenesis, and protection against tissue degeneration.
While BPC-157, TB-500, GHK-Cu, and KPV share overlapping biological themes—namely cytoprotection, inflammation control, and regenerative signaling—they act through distinct molecular pathways. Combining these peptides into a single formulation enables researchers to study how complementary mechanisms converge to influence healing kinetics, tissue quality, and long-term functional outcomes.
This blend was developed to simplify experimental design by reducing the logistical complexity associated with administering multiple peptides independently. By consolidating these peptides into one formulation, researchers may focus on mechanistic interrogation, biomarker analysis, and outcome measurement rather than protocol complexity.
Biochemical Characteristics
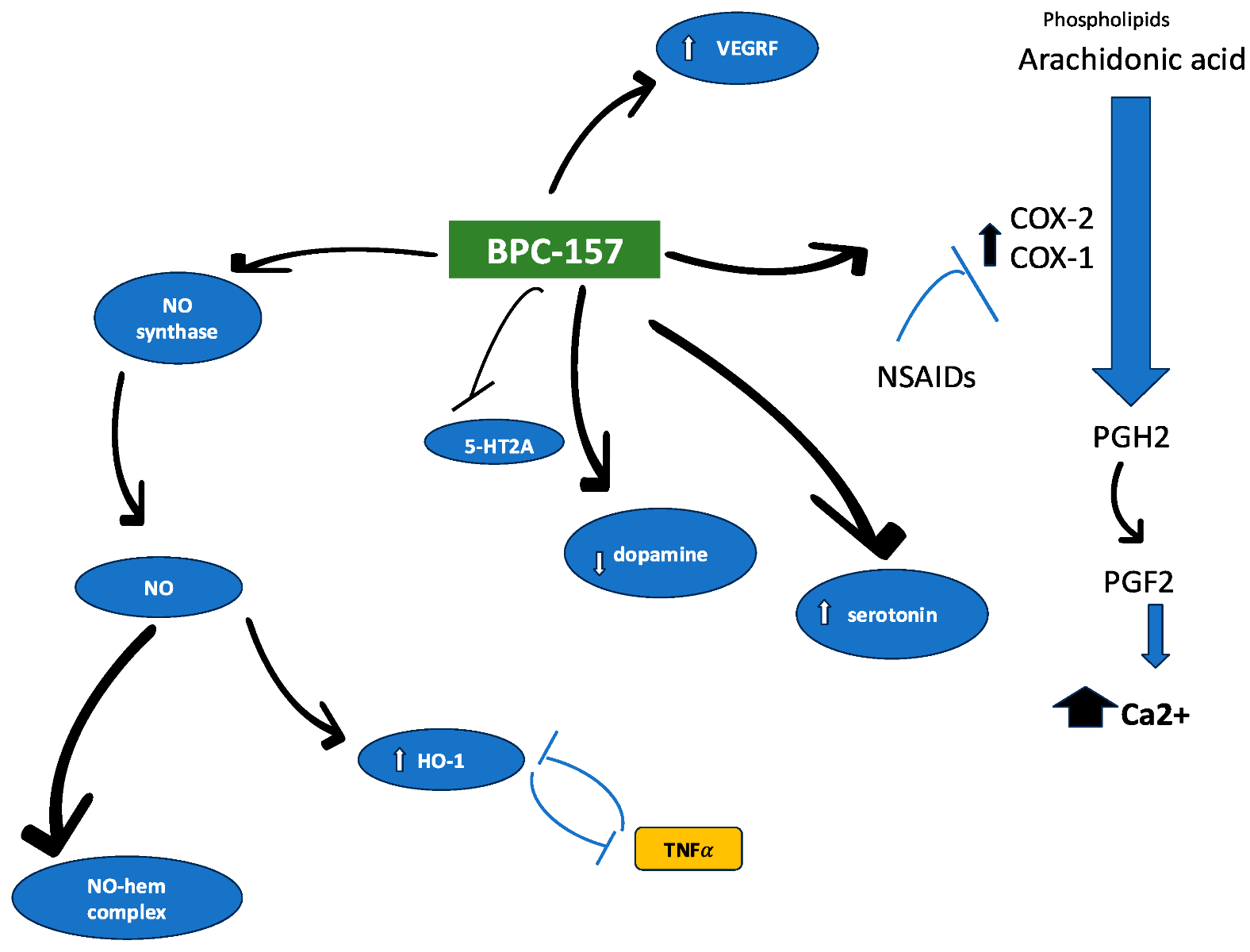
BPC-157 is a synthetic pentadecapeptide derived from a naturally occurring gastric cytoprotective protein known as Body Protective Compound (BPC). It is notable for its stability in gastric environments, rapid systemic distribution, and profound influence on nitric oxide signaling and gene expression related to vascular and immune function.
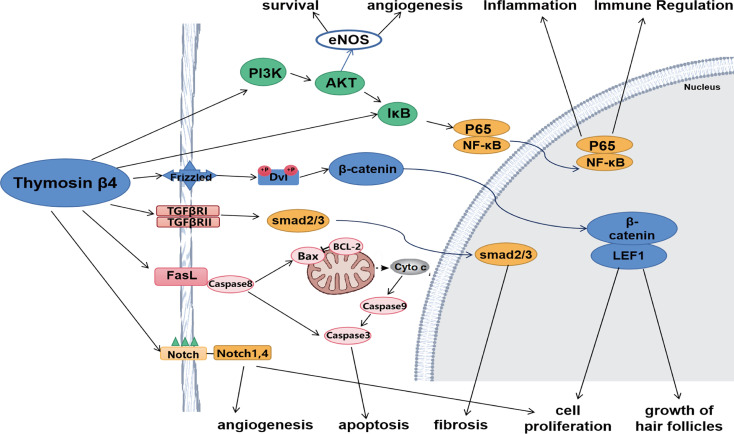
TB-500 is a synthetic peptide based on thymosin beta-4, an actin-binding protein involved in cytoskeletal dynamics, cell migration, and angiogenesis. TB-500 retains both the actin-sequestering properties of thymosin beta-4 and its “moonlighting” function as a regulator of gene expression and inflammatory signaling.
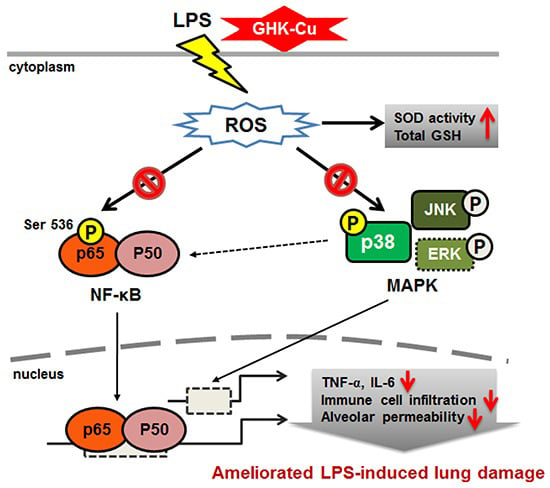
GHK-Cu is a copper-complexed tripeptide (Gly-His-Lys-Cu²⁺) naturally present in human plasma, saliva, and urine. It plays a regulatory role in extracellular matrix remodeling, antioxidant defense, and gene expression related to tissue repair and immune balance.
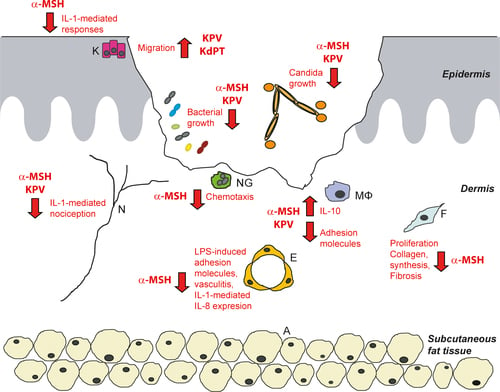
KPV is a tripeptide derived from the C-terminal sequence of α-melanocyte-stimulating hormone (α-MSH). Despite its small size, KPV exhibits potent anti-inflammatory activity and demonstrates selective action in inflamed tissues while largely sparing normal immune responses.
Research Applications
This peptide blend is of interest for research in tissue regeneration, musculoskeletal injury, inflammatory disorders, fibrosis prevention, and aging-associated tissue decline. Investigational models include skin wounds, tendon and ligament injury, muscle degeneration, gastrointestinal inflammation, pulmonary injury, and neuroinflammatory conditions.
The combination allows exploration of how coordinated modulation of nitric oxide signaling, cytokine production, extracellular matrix remodeling, and stem-cell-related pathways influences healing speed, scar quality, and long-term tissue function.
Additional areas of research interest include angiogenesis, immune cell recruitment, oxidative stress mitigation, and regulation of gene expression patterns associated with chronic inflammation and aging.
Pathway / Mechanistic Context
BPC-157 exerts a central role in this blend through its modulation of nitric oxide (NO) signaling. It influences endothelial nitric oxide synthase (eNOS) activity via Src-Caveolin-1 interactions, promoting vascular stability, angiogenesis, and immune regulation. These effects are closely tied to its ability to alter expression of genes such as Egr, Nos, Vegf, and Srf.
TB-500 complements this activity by regulating cytoskeletal dynamics through actin sequestration while simultaneously modulating inflammatory signaling pathways such as NF-κB, Toll-like receptor cascades, PI3K/Akt/eNOS, and TGF-β. These combined actions support cell migration, reduce fibrosis, and enhance tissue regeneration.
GHK-Cu contributes by balancing extracellular matrix turnover through coordinated activation of metalloproteinases and anti-proteases. It also suppresses pro-inflammatory cytokines (TNF-α, IL-6), scavenges reactive oxygen species, and influences gene networks involved in antioxidant defense and tissue remodeling.
KPV provides high-level regulation of inflammation by inhibiting NF-κB signaling, reducing adhesion molecule expression, and suppressing downstream cytokines such as IL-8. Notably, KPV exhibits tissue-selective activity, acting primarily where inflammation is excessive while preserving physiological immune responses necessary for healing.
Source: MDPI
Source: PubMed
Source: PubMed
Source: Wiley Online Library
Preclinical Research Summary
Preclinical studies of BPC-157 demonstrate accelerated wound closure, enhanced angiogenesis, reduced inflammatory infiltration, and improved functional outcomes in tendon, muscle, gastrointestinal, and cardiovascular injury models. TB-500 has shown efficacy in promoting cell migration, reducing fibrosis, and enhancing regeneration in cardiac, muscular, ocular, and dermal injury models.
GHK-Cu has been shown to improve wound healing rates, reduce infection incidence, modulate gene expression associated with tissue regeneration, and protect against oxidative and inflammatory injury in pulmonary, dermal, and neural tissues. KPV has demonstrated broad anti-inflammatory activity across multiple organ systems, with particular efficacy in reducing scarring and pathological inflammation.
Collectively, these peptides demonstrate complementary effects that suggest additive or synergistic outcomes when combined, particularly in models of chronic inflammation, delayed healing, and age-associated tissue degeneration.
Form & Analytical Testing
This blend is supplied as a research-grade peptide formulation intended for in-vitro and preclinical laboratory use. Analytical verification may include identity confirmation, purity assessment, and stability characterization using methods such as mass spectrometry and chromatography, depending on batch-specific documentation.
About The Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Patrycja Kleczkowska, Assoc. Prof. Ph.D. Eng., is an employee of the Military Institute of Hygiene and Epidemiology as a Deputy Director for scientific affairs. As an employee of the Maria Sklodowska-Curie Medical Academy in Warsaw, she deals with didactic activities and scientific work focusing on the field of pharmacology. As an employee of the Polish Academy of Sciences from 2005 to 2015, she began her work on designing and synthesizing drugs with biological activity, mainly in the area of analgesic and neuroprotective effects. Currently, she is expanding her knowledge and skills on the toxicity of new biologically active potential therapeutic substances and defining their safety profiles.
Patrycja Kleczkowska, Assoc. Prof. Ph.D. Eng is being referenced as one of the leading scientists involved in the research and development of BPC-157, TB-500, GHK-Cu and KPV. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Patrycja Kleczkowska, Assoc. Prof. Ph.D. Eng is listed in [3] under the referenced citations.
Referenced Citations
- M.-J. Hsieh et al., “Modulatory effects of BPC 157 on vasomotor tone and the activation of Src-Caveolin-1-endothelial nitric oxide synthase pathway,” Sci Rep, vol. 10, no. 1, p. 17078, Oct. 2020, doi: 10.1038/s41598-020-74022-y.
- L. He et al., “Pharmacokinetics, distribution, metabolism, and excretion of body-protective compound 157, a potential drug for treating various wounds, in rats and dogs,” Front Pharmacol, vol. 13, p. 1026182, Dec. 2022, doi: 10.3389/fphar.2022.1026182.
- M. Józwiak, M. Bauer, W. Kamysz, and P. Kleczkowska, “Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review,” Pharmaceuticals, vol. 18, no. 2, Art. no. 2, Feb. 2025, doi: 10.3390/ph18020185.
- Y. Xing, Y. Ye, H. Zuo, and Y. Li, “Progress on the Function and Application of Thymosin β4,” Front Endocrinol (Lausanne), vol. 12, p. 767785, Dec. 2021, doi: 10.3389/fendo.2021.767785.
- K. Pawar, “Recent Advances in KPV Peptide Delivery,” Journal of Pharmaceutics & Drug Delivery Research, vol. 2022, Apr. 2022, Accessed: Aug. 16, 2025. [Online]. Available: https://www.scitechnol.com/abstract/recent-advances-in-kpv-peptiderndelivery-18216.html
- T. Brzoska, T. A. Luger, C. Maaser, C. Abels, and M. Böhm, “Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases,” Endocr. Rev., vol. 29, no. 5, Art. no. 5, Aug. 2008, doi: 10.1210/er.2007-0027.
- E. Lee, C. Walker, and B. Ayadi, “Effect of BPC-157 on Symptoms in Patients with Interstitial Cystitis: A Pilot Study,” Altern Ther Health Med, vol. 30, no. 10, pp. 12–17, Oct. 2024.
- P. Sikirić et al., “The influence of a novel pentadecapeptide, BPC 157, on N(G)-nitro-L-arginine methylester and L-arginine effects on stomach mucosa integrity and blood pressure,” Eur J Pharmacol, vol. 332, no. 1, pp. 23–33, Jul. 1997, doi: 10.1016/s0014-2999(97)01033-9.
- S. S. Iyer and G. Cheng, “Role of Interleukin 10 Transcriptional Regulation in Inflammation and Autoimmune Disease,” Crit Rev Immunol, vol. 32, no. 1, pp. 23–63, 2012.
- G. Sosne, P. Qiu, and M. Kurpakus-Wheater, “Thymosin beta-4 and the eye: I can see clearly now the pain is gone,” Ann N Y Acad Sci, vol. 1112, pp. 114–122, Sep. 2007, doi: 10.1196/annals.1415.004.
- T. Huang et al., “Body protective compound-157 enhances alkali-burn wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro,” Drug Des Devel Ther, vol. 9, pp. 2485–2499, 2015, doi: 10.2147/DDDT.S82030.
- T. Hara, Y. Nakayama, and N. Nara, “[Regenerative medicine of skeletal muscle],” Rinsho Shinkeigaku, vol. 45, no. 11, pp. 880–882, Nov. 2005.
- Q. Zhang, L. Yan, J. Lu, and X. Zhou, “Glycyl-L-histidyl-L-lysine-Cu2+ attenuates cigarette smoke-induced pulmonary emphysema and inflammation by reducing oxidative stress pathway,” Front Mol Biosci, vol. 9, p. 925700, 2022, doi: 10.3389/fmolb.2022.925700.
- J.-R. Park, H. Lee, S.-I. Kim, and S.-R. Yang, “The tri-peptide GHK-Cu complex ameliorates lipopolysaccharide-induced acute lung injury in mice,” Oncotarget, vol. 7, no. 36, pp. 58405–58417, Sep. 2016, doi: 10.18632/oncotarget.11168.
- M. Kukowska, M. Kukowska-Kaszuba, and K. Dzierzbicka, “In vitro studies of antimicrobial activity of Gly-His-Lys conjugates as potential and promising candidates for therapeutics in skin and tissue infections,” Bioorg Med Chem Lett, vol. 25, no. 3, pp. 542–546, Feb. 2015, doi: 10.1016/j.bmcl.2014.12.029.
- M. Cutuli, S. Cristiani, J. M. Lipton, and A. Catania, “Antimicrobial effects of alpha-MSH peptides,” J Leukoc Biol, vol. 67, no. 2, Art. no. 2, Feb. 2000, doi: 10.1002/jlb.67.2.233.
- P. Sikiric et al., “Stable Gastric Pentadecapeptide BPC 157 as Useful Cytoprotective Peptide Therapy in the Heart Disturbances, Myocardial Infarction, Heart Failure, Pulmonary Hypertension, Arrhythmias, and Thrombosis Presentation,” Biomedicines, vol. 10, no. 11, p. 2696, Oct. 2022, doi: 10.3390/biomedicines10112696.
- L. Pickart and A. Margolina, “Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data,” Int J Mol Sci, vol. 19, no. 7, p. 1987, Jul. 2018, doi: 10.3390/ijms19071987.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







