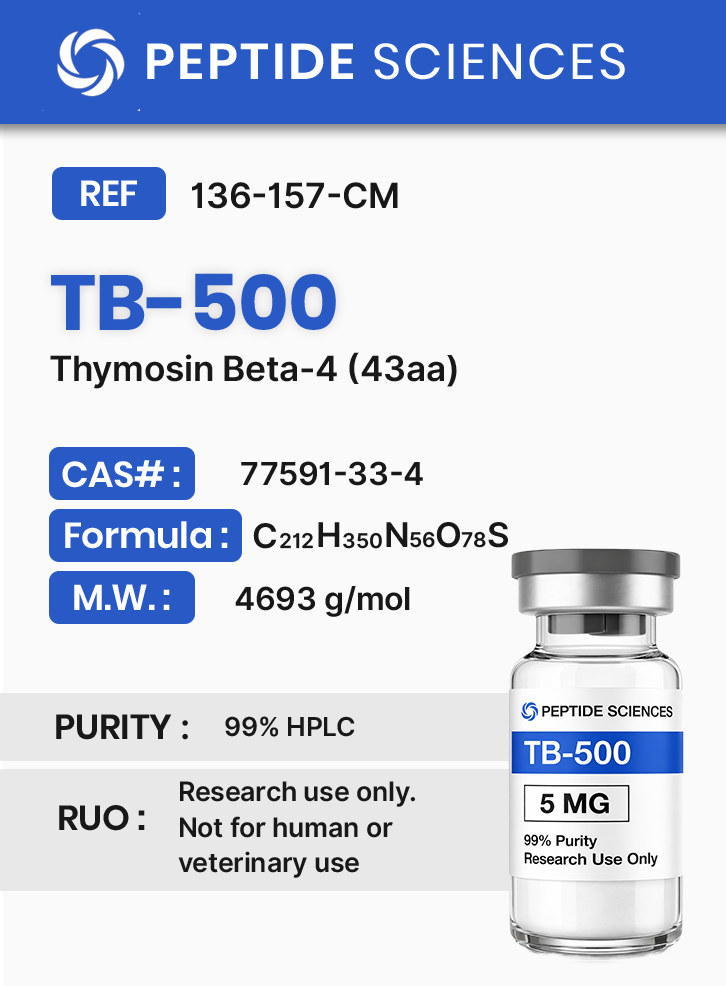
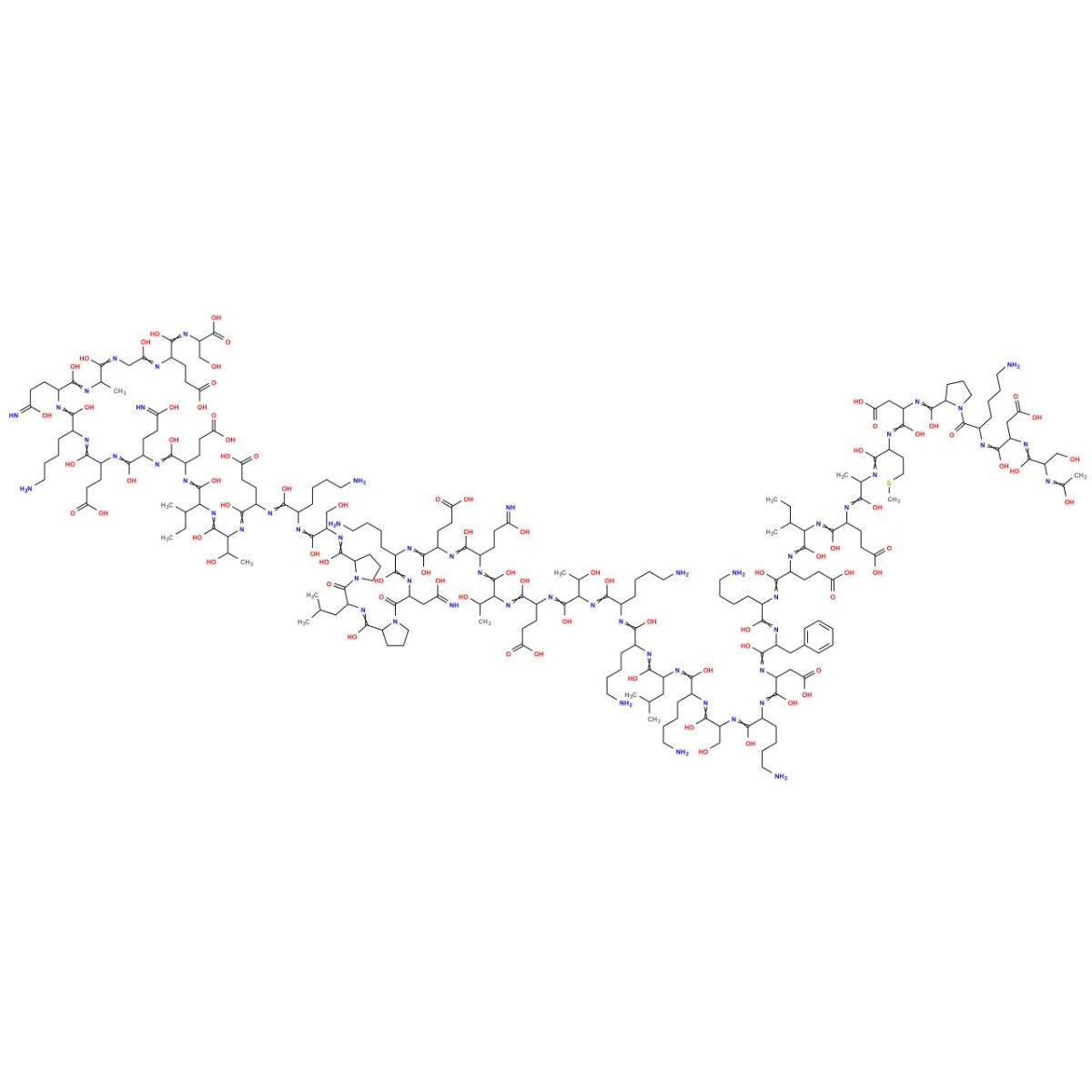
TB-500 (Thymosin Beta-4) (43aa)
$85.00
TB-500 (Thymosin Beta-4) is a research peptide corresponding to a 43–amino acid sequence of Thymosin Beta-4, an actin-binding protein involved in cytoskeletal organization and tissue regeneration. Preclinical studies demonstrate that TB-500 regulates cell migration, angiogenesis, and actin polymerization through modulation of the β-actin/G-actin cycle and related growth factor signaling. It serves as a model compound for studying wound repair, cellular motility, and peptide-mediated regenerative processes.
For research use only. Not for human consumption.
References:
Goldstein AL et al., Ann NY Acad Sci, 2007 1112:148–154
Huff T et al., J Mol Biol, 2001 312(3):593–606
Malinda KM et al., FASEB J, 1999 13(14):2106–2114
Overview
TB-500 is a synthetic 43-amino-acid peptide corresponding to the thymosin beta-4 (Tβ4) sequence motif widely studied in cytoskeletal biology. In experimental systems, thymosin beta peptides are used to interrogate actin monomer (G-actin) sequestration, actin filament (F-actin) dynamics, and downstream processes that depend on cytoskeletal remodeling, including cell migration, neurite outgrowth, endothelial cell behavior, and extracellular matrix (ECM) remodeling. Reported preclinical literature includes cell-based assays and animal studies examining oxidative stress signaling, angiogenic transcriptional programs, epithelial and stromal re-epithelialization kinetics, and tissue remodeling endpoints.
Biochemical Characteristics
Research Applications
TB-500 is used in research workflows to probe cytoskeleton-dependent phenotypes and signaling readouts in controlled laboratory settings. Common experimental contexts include:
- Actin dynamics assays (G-actin sequestration, F-actin polymerization state, stress fiber organization)
- Cell migration and adhesion phenotyping (scratch assays, transwell migration, focal adhesion remodeling)
- Endothelial biology and angiogenic signaling readouts (tube formation assays, VEGF-axis transcriptional endpoints)
- Oxidative stress and innate signaling modules in cell systems (e.g., TLR-adaptor pathway readouts under defined stressors)
- Neural cell support and remodeling paradigms (glial lineage responses, neurite-associated morphology in preclinical models)
- Host-pathogen and adjunct biology paradigms in animal infection models (quantitative microbiology endpoints such as CFUs and inflammatory mediator readouts)
Pathway / Mechanistic Context
Thymosin beta peptides are classically described as actin-binding factors that sequester G-actin, thereby influencing the available monomer pool for filament assembly. Because actin polymerization state regulates membrane protrusion, endocytosis, cytokinesis, and mechanotransduction, modulation of G-actin/F-actin balance can shift cellular behaviors that rely on cytoskeletal remodeling.
Across preclinical systems, TB-4/TB-500-associated readouts have been reported alongside changes in angiogenic transcriptional programs (including VEGF-related signaling), ECM remodeling markers, and stress-response pathways. In certain cell models, thymosin beta 4 has been studied in the context of oxidative-stress signaling and innate pathway nodes (including TLR-adaptor signaling modules), where experimental designs evaluate pathway-linked mediator levels and cell viability markers under defined stress conditions.
Preclinical Research Summary
1. Neural Tissue Models & Support-Cell Biology
In rat spinal cord injury models, thymosin β4 has been evaluated using histological, vascular, and functional/behavioral readouts, with study designs interrogating cellular responses in injured tissue and associated remodeling endpoints[1]. Review literature further discusses thymosin β4 within regenerative biology frameworks for neurological injury models, emphasizing mechanistic hypotheses and preclinical evidence streams rather than clinical translation[2].
In spinal cord–derived neural stem/progenitor cell models, thymosin beta 4 has been studied under oxidative-stress conditions with pathway-level readouts mapped to the TLR4/MyD88 axis, including changes in oxidative mediator levels and viability-associated measures in vitro[3].
2. Vascular Biology, Angiogenic Signaling, and ECM Remodeling
Thymosin β4 has been reported as a modulator of angiogenic programs in preclinical systems, with literature describing relationships to VEGF-associated signaling and vascular development/remodeling endpoints[4]. Proposed mechanistic contexts include cell migration, ECM remodeling, and endothelial/pericyte-associated processes, evaluated using model-specific molecular and histologic readouts.
3. Hair Follicle and Skin-Appendage Model Readouts
Mouse studies involving altered thymosin beta 4 expression have reported differences in hair follicle-associated phenotypes, and thymosin beta 4 has been evaluated for effects on stem-cell migration and differentiation readouts in these experimental contexts[5].
4. Infection Models and Adjunct Biology with Antibiotics
In a mouse model of Pseudomonas aeruginosa-induced keratitis, thymosin beta 4 has been studied in combination with ciprofloxacin, with outcome measures including bacterial colony forming units (CFUs), neutrophil infiltration, and oxidative/inflammatory mediator readouts after defined treatment windows[6].
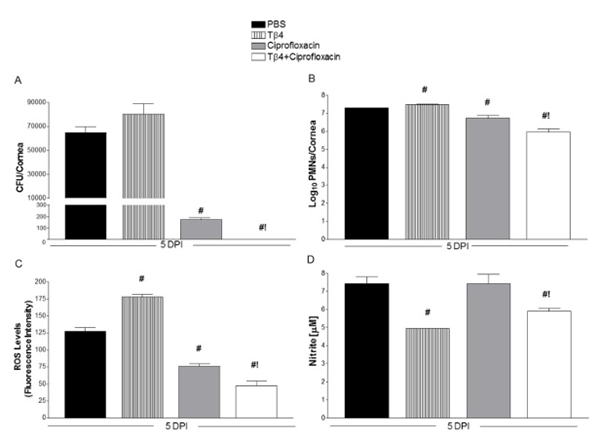 A. Number of colony forming units (CFUs) of bacteria after 5 days of treatment. Note that none are detectable when ciprofloxicin is combined with TB-4.
A. Number of colony forming units (CFUs) of bacteria after 5 days of treatment. Note that none are detectable when ciprofloxicin is combined with TB-4.
B. Shows number of neutrophils in the corneas of treated mice, an indication of inflammation.
C. Measure of reactive oxygen species in corneas of mice after 5 days of treatment.
D. Nitrate levels from corneal lysates.
Source: PubMed
5. Cardiovascular and Renal Model Systems
Literature has examined thymosin β4 and related pathway components in cardiovascular and renal model contexts, including angiogenic remodeling, endothelial migration phenotypes, inflammatory mediator profiles, and fibrosis-associated molecular markers, depending on the model and experimental endpoint selection[7]. Injectable hydrogel formulations incorporating collagen and thymosin β4 have been evaluated in myocardial ischemia-related experimental paradigms with readouts such as angiogenesis-associated markers and epicardial cell migration endpoints in preclinical designs[8].
6. Neurodegeneration-Adjacent Cellular Models and Proteostasis
In HT22 cell experiments using prion peptide (PrP 106–126) exposure, thymosin beta 4 has been studied for effects on autophagy-associated pathways and signaling readouts under defined in vitro conditions[9].
7. Summary
Across preclinical literature, TB-4/TB-500-related experimental work is commonly positioned around cytoskeletal regulation (actin monomer sequestration), migration-associated phenotypes, stress-response signaling, and tissue remodeling endpoints. Interpretation of outcomes is model-dependent and should be evaluated within each study’s dosing regimen, route, timecourse, and assay selection. This material is presented for scientific context and does not imply suitability for any non-laboratory purpose.
Form & Analytical Testing
TB-500 is supplied as a synthetic research peptide intended for controlled laboratory workflows. Researchers commonly document identity and traceability using sequence information and registry identifiers (e.g., CAS and PubChem CID). Standard laboratory qualification may include chromatographic purity assessment (e.g., HPLC-based methods) and mass confirmation (e.g., MS-based methods) as part of internal QC practices.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Allan L. Goldstein, MD, Allan L. Goldstein is professor and Catharine B. & William McCormick Chair of the department of Biochemistry and Molecular Biology at The George Washington University School of Medicine and Health Sciences, where he has served since 1978. Thymosins were discovered in the mid 1960’s, when Allan Goldstein from the Laboratory of Abraham White at the Albert Einstein College of Medicine in New York studied the role of the thymus in development of the vertebrate immune system. He is a world-renowned authority on the thymus gland and the workings of the immune system, and co-discoverer of the thymosins. Dr. Goldstein is the author of over 400 scientific articles in professional journals, the inventor on more than 15 U.S. Patents, and the editor of several books in the fields of biochemistry, biomedicine, immunology and neuro-science. He is on the editorial boards of numerous scientific and medical journals and has been a consultant to many re-search organizations in industry and government; co-founder of The Institute for Advanced Studies in Aging and Geriatric Medicine, a non-profit research and educational institute; a member of the Board of Trustees of the Albert Sabin Vaccine Institute; and serves as the Chairman of the Board of RegeneRx Biopharmaceuticals. Dr. Goldstein received his B.S. from Wagner College in 1959 and his M.S. and Ph.D. from Rutgers University in 1964. He served as a faculty member of the Albert Einstein College of Medicine from 1964 to 1972, and moved to the University of Texas Medical Branch in Galveston in 1972 as professor and director of the division of Biochemistry.
Allan L. Goldstein, MD is being referenced as one of the leading scientists involved in the research and development of TB-500 and other Thymosins. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Goldstein is listed in [11] under the referenced citations.
Referenced Citations
- P. Cheng, F. Kuang, H. Zhang, G. Ju, and J. Wang, “Beneficial effects of thymosin β4 on spinal cord injury in the rat,” Neuropharmacology, vol. 85, pp. 408–416, Oct. 2014. [PubMed]
- M. Chopp and Z. G. Zhang, “Thymosin β4 as a restorative/regenerative therapy for neurological injury and neurodegenerative diseases,” Expert Opin. Biol. Ther., vol. 15 Suppl 1, pp. S9-12, 2015. [PubMed]
- H. Li, Y. Wang, X. Hu, B. Ma, and H. Zhang, “Thymosin beta 4 attenuates oxidative stress-induced injury of spinal cord-derived neural stem/progenitor cells through the TLR4/MyD88 pathway,” Gene, vol. 707, pp. 136–142, May 2019. [PubMed]
- K. N. Dubé and N. Smart, “Thymosin β4 and the vasculature: multiple roles in development, repair and protection against disease,” Expert Opin. Biol. Ther., vol. 18, no. sup1, pp. 131–139, 2018. [PubMed]
- D. Philp, S. St-Surin, H.-J. Cha, H.-S. Moon, H. K. Kleinman, and M. Elkin, “Thymosin beta 4 induces hair growth via stem cell migration and differentiation,” Ann. N. Y. Acad. Sci., vol. 1112, pp. 95–103, Sep. 2007. [PubMed]
- T. W. Carion et al., “Thymosin Beta-4 and Ciprofloxacin Adjunctive Therapy Improves Pseudomonas aeruginosa-Induced Keratitis,” Cells, vol. 7, no. 10, Sep. 2018. [PubMed]
- K. M. Kassem, S. Vaid, H. Peng, S. Sarkar, and N.-E. Rhaleb, “Tβ4-Ac-SDKP pathway: Any relevance for the cardiovascular system?,” Can. J. Physiol. Pharmacol., pp. 1–11, Mar. 2019. [PubMed]
- A. D. Shaghiera, P. Widiyanti, and H. Yusuf, “Synthesis and Characterization of Injectable Hydrogels with Varying Collagen–Chitosan–Thymosin β4 Composition for Myocardial Infarction Therapy,” J. Funct. Biomater., vol. 9, no. 2, Mar. 2018. [PubMed]
- H.-J. Han, S. Kim, and J. Kwon, “Thymosin beta 4-Induced Autophagy Increases Cholinergic Signaling in PrP (106-126)-Treated HT22 Cells,” Neurotox. Res., Dec. 2018. [PubMed]
- Song, Ran & Choi, Hyun & Yang, Hyung-In & Yoo, Myung & Park, Yong-Beom & Kim, Kyoung. (2012). Association between serum thymosin β4 levels of rheumatoid arthritis patients and disease activity and response to therapy. Clinical rheumatology. 31. 1253-8. 10.1007/s10067-012-2011-7. [Research Gate]
- Philp, D., et al. “Thymosin β4 Promotes Angiogenesis, Wound Healing, and Hair Follicle Development.” Mechanisms of Ageing and Development, vol. 125, no. 2, Feb. 2004, pp. 113–115, 10.1016/j.mad.2003.11.005. [PubMed]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.