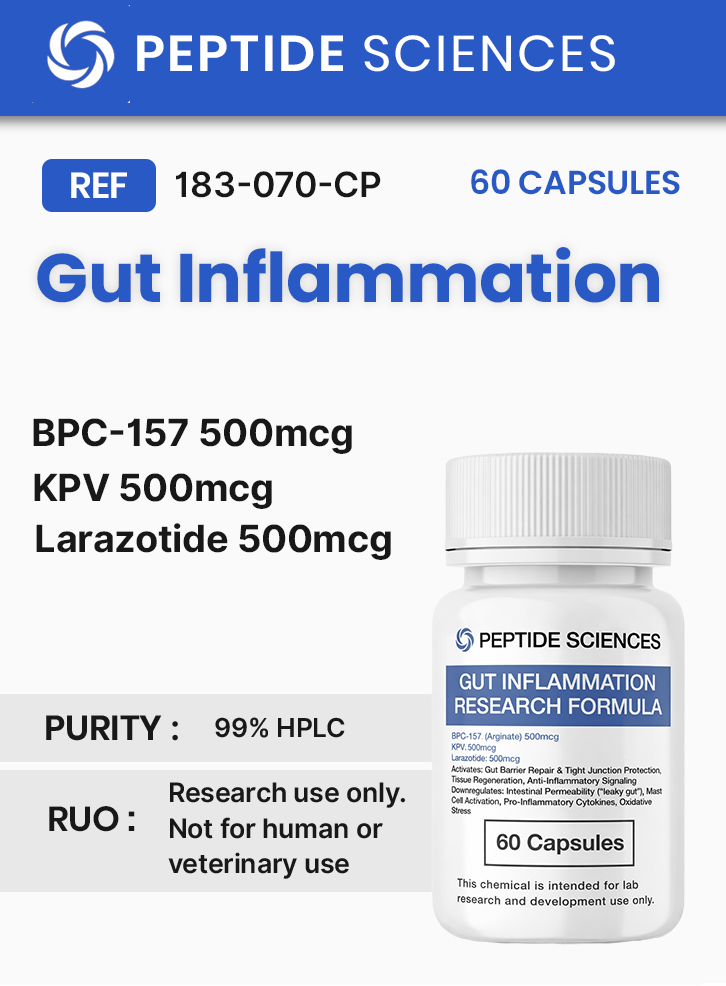
Gut Inflammation (60 Capsules) (Stable BPC-157 Arginate, KPV, Larazotide)
$325.00
Gut Inflammation is a research formulation containing stable BPC-157, KPV, palmitoylethanolamide (PEA), and tributyrin designed for studies on intestinal barrier function and inflammatory modulation. Each component contributes to signaling pathways related to epithelial regeneration, cytokine balance, and microbiome–host interaction. This blend is used in preclinical research investigating mucosal peptide regulation and enteric homeostasis mechanisms.
Across in vitro systems, these peptides are frequently examined using endpoints such as barrier integrity (e.g., transepithelial electrical resistance), permeability tracers, cytokine/chemokine output, and pathway marker changes. Studying them together can support comparative research designs that assess whether combined exposure produces additive or distinct effects on barrier function and inflammatory signaling in experimental gastrointestinal models.
References:
Sikiric P et al., Curr Pharm Des, 2018 24(18):2002–2030
Getting SJ et al., J Immunol, 1999 163(10):5959–5967
Di Paola R et al., Mol Neurobiol, 2018 55(6):4785–4799
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
Overview
This product combines three synthetic peptides—BPC-157 (as an arginate salt), larazotide, and KPV—into a single formulation intended for Research Use Only applications. The blend is designed to support experimental work in gastrointestinal biology where investigators study peptide stability, epithelial barrier regulation, and immune–epithelial signaling under laboratory conditions. BPC-157 is a 15–amino acid peptide sequence widely used in mechanistic research and formulation studies; the arginate salt form is commonly discussed in the context of physicochemical stability and degradation behavior in acidic or variable environmental conditions. Larazotide is an eight–amino acid peptide investigated primarily in intestinal epithelial models to examine tight junction dynamics and barrier modulation. KPV is a tripeptide (Lys–Pro–Val) derived from melanocortin-related sequences and is used in research systems that evaluate inflammatory signaling pathways and mediator output in epithelial and immune cell settings. Current literature largely evaluates these peptides individually; combined-use research is typically framed as a controlled, exploratory design rather than an established, validated outcome.
Biochemical Characteristics
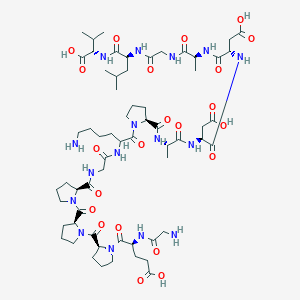
BPC-157
Sequence: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val
Molecular Formula: C62H98N15O22
Molecular Weight: 1419.556 g/mol
PubChem CID: 108101
 Source: PubChem
Source: PubChem
Not all forms of BPC-157 are equivalent in experimental contexts. Some references distinguish between acetate and arginate salt forms and report different stability profiles in simulated gastric acid environments, which may be relevant when designing oral administration studies in laboratory models.
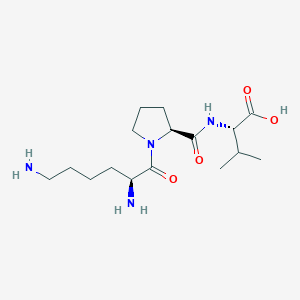
KPV
KPV is a tripeptide analogue derived from the C-terminal region of alpha-melanocyte stimulating hormone (α-MSH) and is investigated for anti-inflammatory and anti-ischemic properties in experimental systems, including gastrointestinal inflammation models.
Amino Acid Sequence: Lys-Pro-Val
Molecular Formula: C16H30N4O4
Molecular Weight: 342.43 g/mol
PubChem CID: 125672
CAS Number: 67727-97-3
Synonyms: MSH (11-13), ACTH(11-13), alpha-MSH(11-13)
 Source: PubChem
Source: PubChem
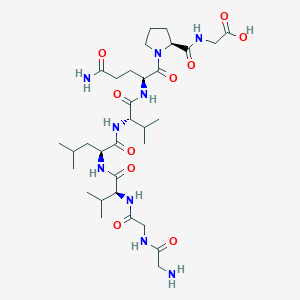
Larazotide
Larazotide is studied as a modulator of intestinal permeability and barrier function, with proposed activity related to regulation of epithelial tight junction dynamics in gastrointestinal models.
Amino Acid Sequence: GGVLVQPG
Molecular Formula: C32H55N9O10
Molecular Weight: 725.845 g/mol
PubChem CID: 9810532
CAS Number: 158818-34-7
Synonyms: AT-1001, AT 2347
 Source: CMGH
Source: CMGH
 Source: PubChem
Source: PubChem
Research Applications
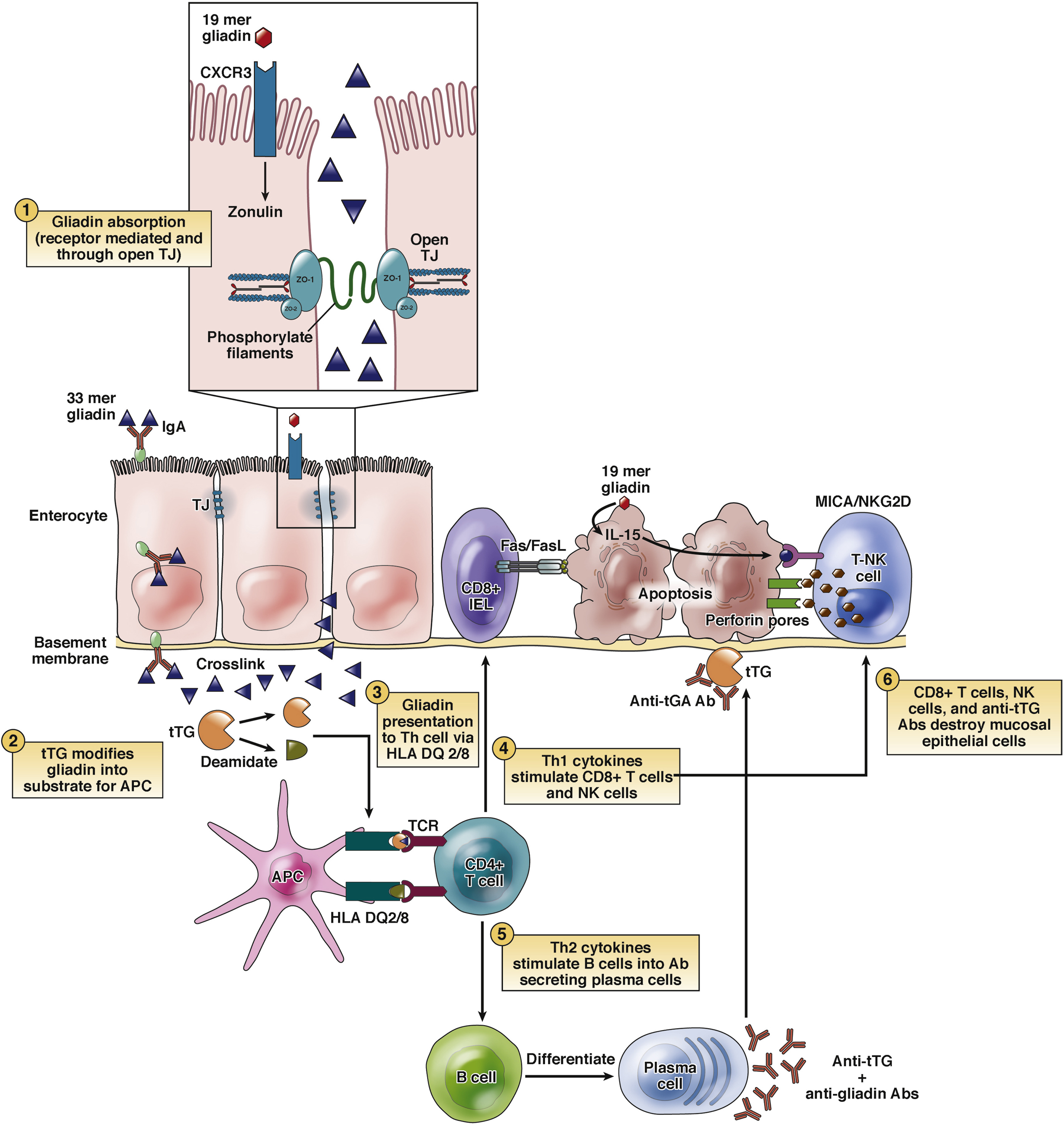
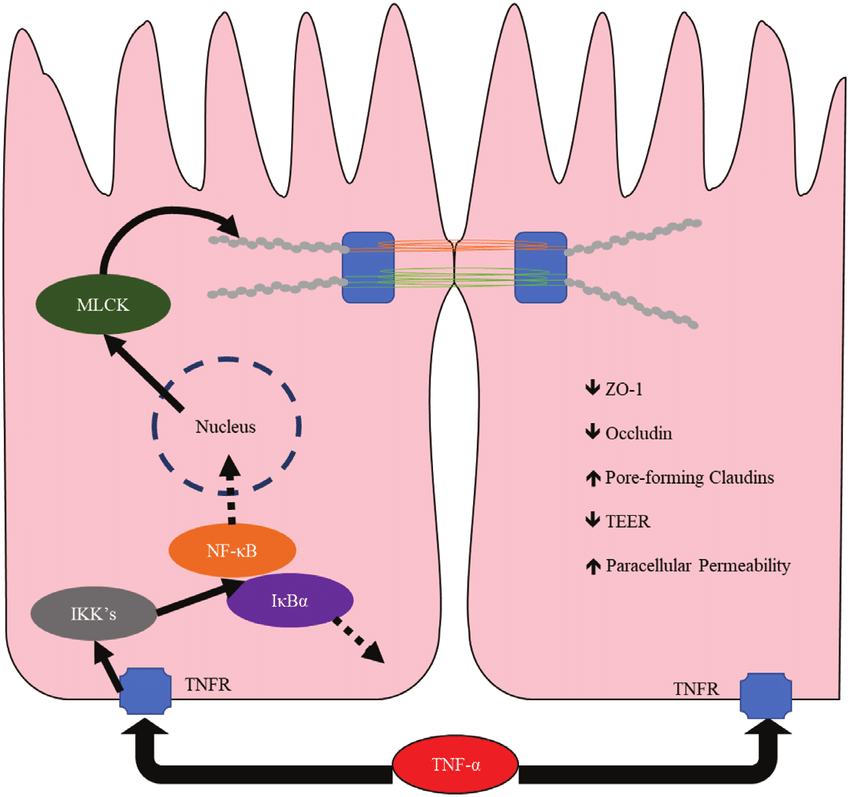
Research on this blend is most appropriately conducted in vitro and/or in animal models, with study designs that quantify measurable endpoints relative to matched controls. In epithelial barrier experiments (e.g., polarized intestinal monolayers), investigators may assess barrier integrity using transepithelial electrical resistance (TEER), permeability tracers (such as fluorescein or dextran conjugates), and localization or expression of tight junction proteins (e.g., occludin, claudins, ZO-1) following exposure to defined stressors. Larazotide-focused work often centers on tight junction state changes and paracellular flux measured over time compared with vehicle controls and, where relevant, positive-control disruptors.
For KPV, common experimental approaches include stimulating epithelial or immune cells with pro-inflammatory triggers and measuring changes in cytokine/chemokine output (e.g., TNF-α, IL-6, IL-8), transcription factor activity, or pathway markers using qPCR, ELISA, Western blotting, or reporter assays. Results are typically interpreted as changes observed under laboratory conditions relative to untreated or vehicle-treated controls, with dose–response and time-course analysis used to differentiate primary effects from downstream shifts.
For BPC-157 arginate, research often includes stability and recovery studies in simulated gastric/intestinal fluids, analytical verification of intact peptide (e.g., HPLC/LC–MS), and comparisons to other salt forms under controlled temperature, pH, or light exposure conditions. In combined protocols, investigators may apply factorial designs (single agents vs. pairwise combinations vs. full blend) to determine whether co-exposure produces additive, non-additive, or distinct changes in barrier metrics and inflammatory signaling endpoints compared with controls. Such studies should maintain clear control groups, predefined readouts, and reproducible analytical methods to support mechanistic interpretation without implying human application.
Pathway / Mechanistic Context
Mechanistically, the proposed synergy is framed as a “layered” approach in experimental GI inflammation models:
- Barrier-first modulation (Larazotide): studied for effects on tight junction regulation and permeability, potentially reducing translocation of luminal antigens that can initiate inflammatory cascades.
- Mucosal protection and repair signaling (BPC-157):
- Inflammatory signal suppression (KPV):
 Source: Research Gate
Source: Research Gate
Preclinical Research Summary
In the published literature, KPV has been evaluated in murine models of inflammatory bowel disease and in formulations designed to target intestinal delivery, with reported reductions in inflammatory markers and improvements in clinical readouts in those experimental settings. Larazotide has been evaluated in barrier-function contexts (including celiac-disease–related research frameworks) and is commonly discussed in relation to tight junction regulation. BPC-157 is discussed across gastrointestinal protection and tissue repair contexts and has also been explored in salt forms that may impact stability in gastric acid environments.
While each peptide has been studied individually, combined three-peptide protocols (BPC-157 + larazotide + KPV) are presented as a hypothesis-generating concept rather than an established, validated regimen in controlled animal or human trials. Accordingly, combination approaches are typically framed as exploratory and intended to inform mechanistic research questions and future experimental design.
Additional discussion in the source material also connects this concept to wound-healing settings, emphasizing experimental themes such as angiogenesis-related pathways, inflammatory cell migration, fibroblast participation in repair, and scar-quality outcomes in preclinical models.
Form & Analytical Testing
If supplied as research peptides, BPC-157, larazotide, and KPV are commonly provided as laboratory materials intended for non-clinical experimentation. Standard analytical characterization for peptide identity and purity may include high-performance liquid chromatography (HPLC) and mass spectrometry (MS), with documentation such as a certificate of analysis (COA) where applicable. Any testing performed and reported should be interpreted as supporting experimental reproducibility rather than clinical performance.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Predrag Sikiric, lead author of “Novel Cytoprotective Mediator, Stable Gastric Pentadecapeptide BPC 157. Vascular Recruitment and Gastrointestinal Tract Healing”, and co-author of “Stable gastric pentadecapeptide BPC 157 in honeybee (Apis mellifera) therapy, to control Nosema ceranae invasions in apiary conditions,” is a Professor of Medical Department at University of Zagreb.
Predrag Sikiric is being referenced as one of the leading scientists involved in the research and development of BPC-157. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Predrag Sikiric is listed in [1] under the referenced citations.
Referenced Citations
- R. Rucman, “Stable pentadecapeptide salts, a process for preparation thereof, a use thereof in the manufacture of pharmaceutical preparations and a use thereof in therapy,” US9850282B2, Dec. 26, 2017.
- M. E. Hiltz and J. M. Lipton, “Antiinflammatory activity of a COOH-terminal fragment of the neuropeptide α-MSH,” FASEB J., vol. 3, no. 11, pp. 2282–2284, 1989, doi: https://doi.org/10.1096/fasebj.3.11.2550304.
- K. Kannengiesser et al., “Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease,” Inflamm. Bowel Dis., vol. 14, no. 3, pp. 324–331, Mar. 2008, doi: 10.1002/ibd.20334.
- B. Xiao et al., “Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis,” Mol. Ther. J. Am. Soc. Gene Ther., vol. 25, no. 7, pp. 1628–1640, Jul. 2017, doi: 10.1016/j.ymthe.2016.11.020.
- M. Cutuli, S. Cristiani, J. M. Lipton, and A. Catania, “Antimicrobial effects of alpha-MSH peptides,” J. Leukoc. Biol., vol. 67, no. 2, pp. 233–239, Feb. 2000, doi: 10.1002/jlb.67.2.233.
- M. F. Masman et al., “Synthesis and conformational analysis of His-Phe-Arg-Trp-NH2 and analogues with antifungal properties,” Bioorg. Med. Chem., vol. 14, no. 22, pp. 7604–7614, Nov. 2006, doi: 10.1016/j.bmc.2006.07.007.
- J. Shi, M. Barakat, D. Chen, and L. Chen, “Bicellular Tight Junctions and Wound Healing,” Int. J. Mol. Sci., vol. 19, no. 12, Dec. 2018, doi: 10.3390/ijms19123862.
- M. Schiller et al., “Human Dermal Fibroblasts Express Prohormone Convertases 1 and 2 and Produce Proopiomelanocortin-Derived Peptides,” J. Invest. Dermatol., vol. 117, no. 2, pp. 227–235, Aug. 2001, doi: 10.1046/j.0022-202x.2001.01412.x.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.