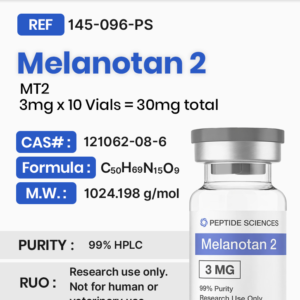
Bronchogen 20mg (Bioregulator)
$70.00
Bronchogen is a short bioregulatory peptide (Ala–Glu–Asp–Leu) studied in laboratory and preclinical models for its tissue-directed activity in lung-related systems. In controlled research settings, Bronchogen is evaluated for how it may influence gene transcription programs, epithelial integrity markers, and inflammatory signaling pathways associated with airway and alveolar tissue function.
Experimental studies in vitro and in animal models commonly assess Bronchogen using endpoints such as bronchial epithelium morphology, cytokine expression profiles, and surfactant-related functional markers. Additional investigations explore its interaction with DNA stability metrics and age-associated transcriptional changes, positioning Bronchogen as a mechanistic research tool for studying cellular regulation, tissue maintenance, and senescence-linked pathways in lung tissue models.
For research use only. Not for human consumption.
References:
Khavinson VK et al., Bull Exp Biol Med, 2004 137(5):597–600
Trofimova SV et al., Adv Gerontol, 2015 28(3):432–439
Linkova NS et al., Biochemistry (Moscow), 2012 77(7):874–881
Overview
Bronchogen (Ala–Glu–Asp–Leu; AEDL) is a four–amino acid peptide studied as a bioregulator with reported tissue-preferential activity in lung-associated experimental systems. Within the research literature, Bronchogen is frequently discussed in the context of DNA interaction/stability assays and transcriptional regulation, with investigators using these endpoints to understand how short peptides may modulate cell-state programs relevant to differentiation, tissue maintenance, and inflammatory balance.
In preclinical and laboratory models, Bronchogen is explored as a mechanistic probe for pathways tied to bronchial epithelial structure and function. These studies typically quantify outcomes such as epithelial cell integrity, remodeling-related histology, inflammatory mediator levels, and markers associated with lung surface-tension regulation (often operationalized through surfactant-related measurements in model systems). Findings remain dependent on model design and measurement methods, and should be interpreted strictly as experimental and mechanistic.
Biochemical Characteristics
Amino Acid Sequence: Ala-Glu-Asp-Leu (AEDL)
Chemical Formula: C18H30N4O9
Molecular Mass: 446.45 g/mol
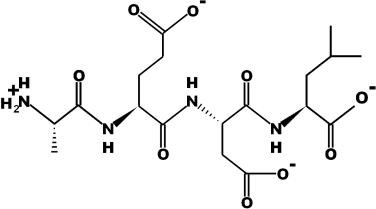 Source: Pepdraw
Source: Pepdraw
As a low-molecular-weight peptide (tetrapeptide), Bronchogen is frequently evaluated in reductionist biochemical systems where sequence-defined ligands can alter macromolecular stability, chromatin-associated binding equilibria, or transcriptional responsiveness. Literature characterizes Bronchogen as a short regulatory peptide with reported effects on DNA thermal transitions under controlled in vitro conditions[1].
Research Applications
Bronchogen is utilized in cell culture, organotypic tissue models, and animal studies to investigate transcriptional regulation and tissue-specific cellular responses. Research applications commonly include DNA thermostability assays, gene-expression analysis, and transcription-factor profiling to assess how Bronchogen exposure may influence differentiation and inflammatory signaling cascades in lung-associated cells.
In preclinical lung pathology models, Bronchogen has been examined using histologic analysis of bronchial epithelium, cytokine quantification, and morphofunctional assessments related to surfactant biology and tissue remodeling. Aging-focused experimental designs further explore whether Bronchogen modulates transcriptional patterns linked to cellular senescence in lung tissue, positioning the peptide as a mechanistic probe for studying regulatory pathways under controlled laboratory conditions.
Pathway / Mechanistic Context
Published preclinical work has positioned Bronchogen within a broader class of short peptides investigated for their capacity to influence gene expression and differentiation programs in a tissue-biased manner. Mechanistic interpretations in the literature include:
- DNA stability modulation: In vitro calorimetry has reported an increase in DNA melting temperature in the presence of Bronchogen, consistent with altered DNA duplex stability or changes in hydration/ionic microenvironment around nucleic acids[1]. Such effects are commonly interrogated via thermal denaturation curves and calorimetric enthalpy/entropy estimates.
- Transcriptional network responsiveness: Organotypic and cellular studies of peptide bioregulators have reported changes in differentiation-related outputs with tissue specificity and age-context dependence, including associations with CXCL12- and HOX-family regulatory signatures in experimental systems[2],[3]. These nodes intersect with chemokine-driven migration cues and HOX-linked patterning/differentiation programs, respectively, and are frequently studied via transcriptomics and transcription factor activity assays.
- Inflammation-linked signaling readouts in animal models: In rodent obstructive lung pathology models, peptide-associated shifts in epithelial morphofunctional parameters and inflammatory marker profiles have been described in the cited literature[5],[6]. These readouts are typically interpreted through the lens of cytokine networks, epithelial barrier state, and remodeling-associated gene expression programs.
- Cross-kingdom gene regulatory effects: In plant systems, short exogenous peptides have been reported to regulate expression of developmental gene families (CLE, KNOX1, GRF), supporting use as tools to probe conserved features of peptide-mediated transcriptional control and developmental signaling[7].
Preclinical Research Summary
DNA thermostability (in vitro): Microcalorimetric evaluation has reported increased DNA thermostability in the presence of Bronchogen, reflected as an elevation in melting temperature under the experimental conditions described[1]. This observation is used experimentally as a proxy measure for peptide-associated shifts in DNA duplex stability and can be integrated into mechanistic studies of DNA–ligand interactions and nucleic-acid biophysics.
Tissue-specific and age-context differentiation effects (preclinical culture models): Studies in organotypic tissue culture and cell systems have described tissue-biased effects of synthetic short peptides on differentiation-related outputs, including comparative observations across young and older experimental contexts[2],[3]. These reports support use of Bronchogen-class peptides as tools for probing transcriptional and differentiation dynamics in controlled model systems.
Rodent obstructive lung pathology models (in vivo): Preclinical rodent studies cited below have evaluated peptide-associated changes in bronchial epithelium morphofunctional parameters and inflammation-linked readouts in induced obstructive lung pathology models[5],[6]. These reports provide an experimental basis for laboratory investigation of epithelial remodeling, cytokine signaling, and barrier-state endpoints within animal model frameworks.
Plant gene expression regulation (in vivo plant model): In Nicotiana tabacum, short exogenous peptides have been reported to regulate expression of CLE, KNOX1, and GRF family genes, providing a platform for mechanistic studies of peptide-driven transcriptional regulation in plant developmental biology[7].
Form & Analytical Testing
Bronchogen is supplied as a synthetic research peptide intended for laboratory workflows. Typical analytical characterization for peptides of this class may include (as applicable to a given lot/specification):
- Identity confirmation by mass spectrometry (MS)
- Purity assessment by HPLC/UPLC
- Optional residual solvent and counterion testing depending on synthesis and finishing processes
Researchers should design handling, storage, and reconstitution protocols according to institutional laboratory SOPs and the specific analytical documentation accompanying the material (e.g., COA where provided).
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
 Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Prof. Vladimir Khavinson is being referenced as one of the leading scientists involved in the research and development of Bronchogen. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide.
Referenced Citations
- J. R. Monaselidze et al., “Effect of the peptide bronchogen (Ala-Asp-Glu-Leu) on DNA thermostability,” Bull. Exp. Biol. Med., vol. 150, no. 3, pp. 375–377, Jan. 2011, doi: 10.1007/s10517-011-1146-x.
- A. N. Zakutskiĭ, N. I. Chalisova, G. A. Ryzhak, A. I. Aniskina, S. V. Filippov, and P. N. Zeziulin, “[The tissue-specific effect of synthetic peptides-biologic regulators in organotypic tissues culture in young and old rats],” Adv. Gerontol. Uspekhi Gerontol., vol. 19, pp. 93–96, 2006.
- V. K. Khavinson, N. S. Linkova, V. O. Polyakova, O. V. Kheifets, S. I. Tarnovskaya, and I. M. Kvetnoy, “Peptides tissue-specifically stimulate cell differentiation during their aging,” Bull. Exp. Biol. Med., vol. 153, no. 1, pp. 148–151, May 2012, doi: 10.1007/s10517-012-1664-1.
- J. Monaselidze, M. Kiladze, M. Gorgoshidze, D. Khachidze, and E. Lomidze, “Influence of anticarcinogenic metalloporphyrin Cu(II)TOEPyP(4) on DNA thermostability in vitro,” Georgian Med. News, no. 175, pp. 57–59, Oct. 2009.
- N. A. Kuzubova, E. S. Lebedeva, I. V. Dvorakovskaya, E. A. Surkova, I. S. Platonova, and O. N. Titova, “Modulating Effect of Peptide Therapy on the Morphofunctional State of Bronchial Epithelium in Rats with Obstructive Lung Pathology,” Bull. Exp. Biol. Med., vol. 159, no. 5, pp. 685–688, Sep. 2015, doi: 10.1007/s10517-015-3047-x.
- O. N. Titova, N. A. Kuzubova, E. S. Lebedeva, T. N. Preobrazhenskaya, E. A. Surkova, and I. V. Dvorakovskaya, “[ANTIINFLAMMATORY AND REGENERATIVE EFFECT OF PEPTIDE THERAPY IN THE MODEL OF OBSTRUCTIVE LUNG PATHOLOGY],” Ross. Fiziol. Zh. Im. I M Sechenova, vol. 103, no. 2, pp. 201–208, Feb. 2017.
- L. I. Fedoreyeva et al., “Short Exogenous Peptides Regulate Expression of CLE, KNOX1, and GRF Family Genes in Nicotiana tabacum,” Biochem. Biokhimiia, vol. 82, no. 4, pp. 521–528, Apr. 2017, doi: 10.1134/S0006297917040149.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.