Vesugen 20mg (Bioregulator)
$65.00
Vesugen is a synthetic short peptide bioregulator derived from vascular tissue extracts, composed of amino acid sequences that influence endothelial function and vascular gene expression. Preclinical studies indicate its role in modulating angiogenic signaling, extracellular matrix remodeling, and smooth muscle cell activity. Vesugen is used in research investigating peptide-mediated vascular homeostasis, endothelial integrity, and microcirculatory regulation mechanisms.
For research use only. Not for human consumption.
References:
Khavinson VK et al., Bull Exp Biol Med, 2003;136(1):45–48
Linkova NS et al., Adv Gerontol, 2011;24(4):628–634
Anisimov VN et al., Biogerontology, 2010;11(2):139–149
Overview
Vesugen (Lys–Glu–Asp; KED) is a synthetic tripeptide investigated as a molecular regulator of vascular-associated gene expression and cellular signaling pathways. Preclinical studies have examined its role in endothelial cell regulation, neuronal cell survival signaling, and transcriptional control mechanisms relevant to vascularized tissues. Due to its short amino acid sequence and linear structure, Vesugen is utilized in controlled laboratory experiments focused on peptide-mediated modulation of cellular and molecular processes.
This compound is supplied exclusively as a laboratory research reagent for use in non-clinical experimental systems, including in-vitro models and in-vivo animal studies.
Biochemical Characteristics
Amino Acid Sequence: Lys–Glu–Asp (KED)
Molecular Formula: C15H26N4O8
Molecular Weight: 390.39 g/mol
PubChem CID: 87571363
Synonyms: lysylglutamyl aspartic acid, SCHEMBL3767701, CHEBI:159909
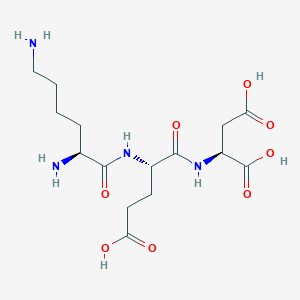 Source: PubChem
Source: PubChem
The tripeptide does not adopt higher-order secondary or tertiary structures under standard laboratory conditions. This structural simplicity facilitates reproducible biochemical interaction studies involving nucleic acids, transcription factors, and intracellular signaling complexes.
Research Applications
Vesugen is employed in experimental research to investigate peptide-driven regulation of endothelial function, neuronal gene expression, and metabolic signaling pathways. Its biochemical properties make it suitable for mechanistic studies examining oxidative stress responses, transcriptional regulation, and peptide-mediated modulation of intracellular enzymes in preclinical models.
- Endothelial cell proliferation and gene expression studies
- Neuronal differentiation and survival signaling research
- Sirtuin-associated metabolic pathway investigations
- Oxidative stress and hypoxia-response models
- Epigenetic regulation and transcriptional control assays
Pathway / Mechanistic Context
Preclinical research indicates that Vesugen influences gene expression through peptide–DNA and peptide–protein interactions that alter transcriptional activity in vascular and neural cell populations. Experimental models have demonstrated modulation of pathways involving sirtuin 1 (SIRT1), endothelin signaling, and oxidative stress response enzymes.
At the molecular level, KED peptides have been associated with regulation of transcription factors involved in apoptosis control, cellular proliferation, and metabolic homeostasis. These effects are studied within controlled laboratory environments using cellular systems and animal models.
Preclinical Research Summary
In-vitro and in-vivo animal studies have examined Vesugen in the context of endothelial regulation, neuronal marker expression, oxidative stress modulation, and metabolic signaling. Rodent-based models have been utilized to explore its effects on vascular integrity, lipid-associated pathways, and transcriptional regulation of cellular survival mechanisms.
Additional investigations have evaluated Vesugen within experimental neurobiology and metabolic research frameworks, focusing on hypoxia-response pathways, mitochondrial regulation, and epigenetic control of gene expression. All findings remain confined to exploratory preclinical research.
Form & Analytical Testing
Vesugen is supplied as a laboratory-grade peptide for analytical and experimental applications. Identity and purity may be confirmed using standard analytical methodologies such as high-performance liquid chromatography (HPLC), mass spectrometry (MS), and amino acid composition analysis.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
 Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Prof. Vladimir Khavinson is being referenced as one of the leading scientists involved in the research and development of Vesugen. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide.
Referenced Citations
- V. K. Khavinson, N. S. Lin’kova, and R. S. Umnov, “Peptide KED: Molecular-Genetic Aspects of Neurogenesis Regulation in Alzheimer’s Disease,” Bull. Exp. Biol. Med., vol. 171, no. 2, pp. 190–193, May 2021, doi: 10.1007/s10517-021-05192-6.
- L. S. Kozina, “[Investigation of antihypoxic properties of short peptides],” Adv. Gerontol. Uspekhi Gerontol., vol. 21, no. 1, pp. 61–67, 2008.
- K. L. Kozlov et al., “[Molecular aspects of vasoprotective peptide KED activity during atherosclerosis and restenosis],” Adv. Gerontol. Uspekhi Gerontol., vol. 29, no. 4, pp. 646–650, 2016.
- L. S. Kozina, A. V. Arutiunian, S. L. Stvolinskiĭ, and V. K. Khavinson, “[Biological activity of regulatory peptides in model experiments in vitro],” Adv. Gerontol. Uspekhi Gerontol., vol. 21, no. 1, pp. 68–73, 2008.
- V. K. Khavinson, S. I. Tarnovskaia, N. S. Lin’kova, E. O. Guton, and E. V. Elashkina, “[Epigenetic aspects of peptidergic regulation of vascular endothelial cell proliferation during aging],” Adv. Gerontol. Uspekhi Gerontol., vol. 27, no. 1, pp. 108–114, 2014.
- C. Sun et al., “SIRT1 improves insulin sensitivity under insulin-resistant conditions by repressing PTP1B,” Cell Metab., vol. 6, no. 4, pp. 307–319, Oct. 2007, doi: 10.1016/j.cmet.2007.08.014.
- S. Nemoto, M. M. Fergusson, and T. Finkel, “SIRT1 functionally interacts with the metabolic regulator and transcriptional coactivator PGC-1{alpha},” J. Biol. Chem., vol. 280, no. 16, pp. 16456–16460, Apr. 2005, doi: 10.1074/jbc.M501485200.
- M. Lagouge et al., “Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha,” Cell, vol. 127, no. 6, pp. 1109–1122, Dec. 2006, doi: 10.1016/j.cell.2006.11.013.
- I. H. Lee et al., “A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy,” Proc. Natl. Acad. Sci. U. S. A., vol. 105, no. 9, pp. 3374–3379, Mar. 2008, doi: 10.1073/pnas.0712145105.
- V. N. Meshchaninov, E. L. Tkachenko, S. V. Zharkov, I. V. Gavrilov, and I. E. Katyreva, “[EFFECT OF SYNTHETIC PEPTIDES ON AGING OF PATIENTS WITH CHRONIC POLYMORBIDITY AND ORGANIC BRAIN SYNDROME OF THE CENTRAL NERVOUS SYSTEM IN REMISSION],” Adv. Gerontol. Uspekhi Gerontol., vol. 28, no. 1, pp. 62–67, 2015.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use







