GHK-Cu (Copper Peptide) (Topical)
$200.00
GHK-Cu (copper tripeptide) (Topical), is a naturally occurring copper–peptide complex composed of glycyl-histidyl-lysine bound to copper (Cu2+) and is studied for its role in cellular stress-response and tissue remodeling pathways. GHK-Cu peptide research commonly examines how this small copper-chelating molecule influences gene-expression programs associated with extracellular matrix organization, oxidative-stress response, and inflammatory signaling under controlled laboratory conditions.
In vitro systems and animal models have evaluated endpoints such as collagen- and elastin-related gene expression, fibroblast activity markers, cytokine signaling profiles including IL-6 and TNF-associated pathways, antioxidant-response genes, and proteasome-related transcriptional changes relative to controls. Major research areas include skin biology models, redox-regulation studies, and extracellular matrix remodeling assays. These biological systems are central to laboratory research because copper homeostasis and matrix regulation are fundamental to cellular adaptation and structural maintenance processes in preclinical environments.
For research use only. Not for human consumption.
References:
Pickart L et al., BioMed Res Int, 2015
Maquart FX et al., FEBS Lett, 1988;238(2):343–346
Siméon A et al., J Invest Dermatol, 2000;115(6):962–968
Overview
GHK-Cu is a copper-binding form of the endogenous tripeptide GHK (glycyl-histidyl-lysine) and is classified as a small regulatory peptide–metal complex studied in molecular and cellular biology research. It was originally identified in human plasma and has since been investigated for its interactions with copper-dependent enzymatic systems and gene-regulatory pathways.
In laboratory settings, GHK-Cu is examined for its association with transcriptional modulation across pathways involved in extracellular matrix dynamics, inflammatory mediator signaling, oxidative balance, and cellular stress adaptation. Because copper functions as a redox-active cofactor in numerous enzymatic reactions, complexation with GHK provides a model for studying controlled copper delivery and its downstream molecular effects. Research frequently compares GHK-Cu to peptide-alone and copper-alone controls to isolate metal-dependent signaling influences within defined experimental systems.
Biochemical Characteristics
Peptide Sequence: Gly-His-Lys (Cu2+)
Molecular Class: Copper-complexed tripeptide
Metal Coordination: Cu2+ bound via histidine and amine residues
GHK exhibits strong affinity for copper ions, and copper coordination can support distinct biochemical behavior compared to the non-complexed tripeptide. In research settings, this copper-binding property is used to explore metal-dependent regulation of ECM synthesis and remodeling, as well as copper-sensitive oxidative stress and signaling pathways. [1]
Research Applications
GHK-Cu is studied in vitro and in animal models to evaluate measurable molecular and cellular changes relative to appropriate control groups. Common endpoints include extracellular matrix–associated gene expression (such as collagen, elastin, and decorin targets), fibroblast proliferation markers, cytokine signaling profiles (including IL-6, TNF, and NF-κB–associated pathways), and oxidative-stress biomarkers.
Transcriptomic studies have reported broad shifts in gene-expression patterns under controlled exposure conditions, including modulation of antioxidant-response genes, DNA-repair–associated markers, and ubiquitin–proteasome system components. In preclinical tissue models, researchers may assess histological remodeling markers, vascularization indicators, and inflammatory mediator expression compared to untreated or vehicle-treated controls.
All reported findings are interpreted within the parameters of the specific experimental design, including model type, concentration, duration, and assay methodology. These observations are confined to laboratory and preclinical research contexts and do not imply clinical application or therapeutic use.
Pathway / Mechanistic Context
Mechanistic research on GHK-Cu commonly explores pathway-level effects related to ECM remodeling and cell–matrix communication, including signaling environments associated with matrix synthesis and organization. Studies have also discussed gene-expression modulation across panels relevant to inflammatory signaling, angiogenic regulators, cell-cycle control, and structural matrix proteins in preclinical and in-vitro systems. [1]
In fibroblast-focused models, publications have reported ECM-associated readouts consistent with collagen and matrix component regulation in culture systems, supporting use of GHK-Cu as a laboratory probe in matrix biology research. [2], [3]
Preclinical Research Summary
Preclinical investigations of GHK-Cu include studies in cultured fibroblasts and skin-relevant cellular systems, with reported outcomes focusing on ECM-related biosynthesis and remodeling markers. Additional research discussions have summarized broader gene-expression and signaling effects in experimental models. All findings are limited to laboratory and experimental research settings and are not indicative of clinical performance. [1]
Form & Analytical Testing
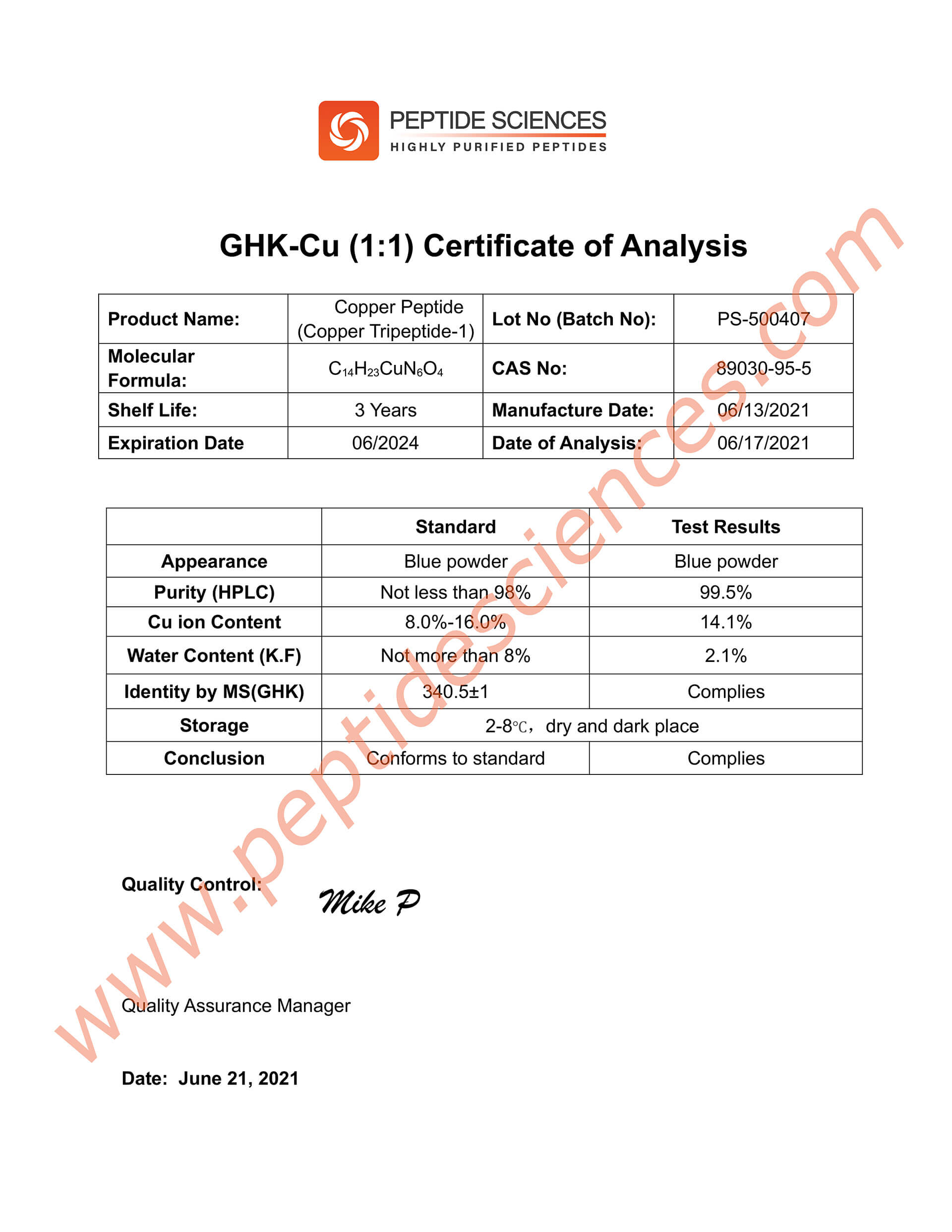
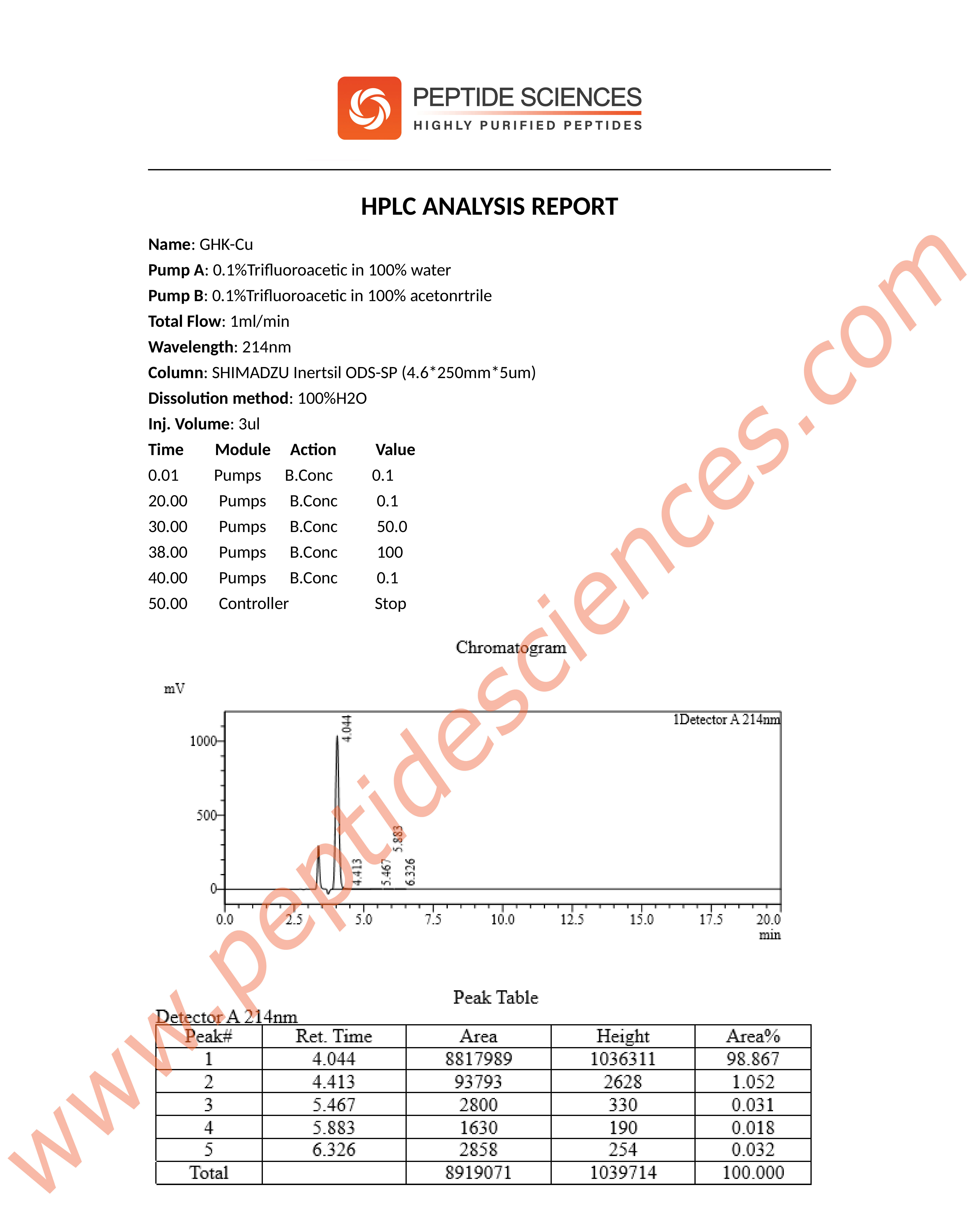
Product identity and purity are commonly verified using established analytical techniques such as HPLC and mass spectrometry (MS), supporting batch-to-batch consistency for research reproducibility.
Quality Documentation
The following quality documentation is provided to support product identity, purity, and analytical verification for laboratory research use. All analyses are batch-specific and supplied for informational and reproducibility purposes only.
Certificate of Analysis (COA)
High Performance Liquid Chromatography (HPLC)
Mass Spectrometry (MS)
Referenced Citations
- Pickart L et al. BioMed Research International. 2015.
- Maquart FX et al. FEBS Letters. 1988;238(2):343–346.
- Siméon A et al. Journal of Investigative Dermatology. 2000;115(6):962–968.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat, or cure any medical condition, ailment, or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.