BPC-157
$100.00
BPC-157 is a synthetic pentadecapeptide (15 amino acids) originally identified as a fragment sequence associated with larger gastric-derived “body protection compound” research. In laboratory settings, BPC-157 is studied as a research peptide for exploring cellular signaling involved in tissue response to injury, including pathways related to fibroblast activity, extracellular matrix (ECM) organization, and endothelial cell behavior.
Preclinical and in vitro studies commonly evaluate BPC-157 in models that measure cell migration, cell survival, angiogenesis-related signaling, and oxidative stress biomarkers under controlled conditions. Because these endpoints are quantifiable in cell culture systems and animal models, BPC-157 is frequently used to probe mechanisms of tissue repair biology, vascular recruitment/collateralization, and inflammation-linked signaling cascades, without implying human outcomes.
For research use only. Not for human consumption.
References:
Sikiric P et al., Curr Pharm Des, 2018;24(18):2002–2030
Kang EA et al., Dig Dis Sci, 2013;58:3310–3317
Seiwerth S et al., Biomedicines, 2018;6(3):75
Overview
BPC-157 (Body Protection Compound-157) is a synthetic peptide composed of 15 amino acids and is studied in research as a fragment associated with larger gastric-derived cytoprotective protein/peptide systems. Within experimental literature, BPC-157 is generally evaluated for mechanistic effects on cellular processes that support barrier integrity, tissue response to injury, and vascular adaptation in controlled models.
Across in vitro assays and animal studies, BPC-157 research commonly focuses on fibroblast-related biology (migration, proliferation, and cytoskeletal organization), endothelial cell signaling relevant to angiogenesis, and broader pathways involving nitric oxide–linked signaling and oxidative stress markers. These studies use defined endpoints such as collagen/ECM deposition metrics, endothelial proliferation assays, and biochemical markers of stress/inflammation to characterize how BPC-157 influences repair-associated pathways at the cellular and tissue level.
Biochemical Characteristics
 Source: PubChem
Source: PubChem
Sequence: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val
Molecular Formula: C62H98N16O22
Molecular Weight: 1419.556 g/mol
PubChem CID: 108101
BPC-157 is a linear peptide lacking disulfide bonds. Its proline-rich sequence contributes to conformational rigidity and reduced susceptibility to proteolytic degradation, supporting stability in aqueous experimental systems.
Research Applications
BPC-157 is used in cell culture, ex vivo, and animal model research to investigate tissue repair mechanisms and vascular recruitment processes. Typical experimental endpoints include fibroblast proliferation/migration assays, wound-closure or scratch assays, cytoskeletal organization readouts (e.g., F-actin–related staining), and signaling pathway markers associated with cell adhesion and motility (often evaluated via phosphorylation-state changes or protein expression panels in controlled systems).
A major research application involves angiogenesis and vascular adaptation studies. Researchers may quantify endothelial cell proliferation and tube formation in vitro, measure pro-angiogenic receptor signaling (including pathways discussed in the literature around VEGFR2-associated activity), and evaluate collateral vessel development in ischemia or occlusion models. These investigations often pair histologic analysis with functional perfusion or tissue viability readouts to understand vascular “recruitment” and restoration of blood flow in model organisms.
Additional research use-cases evaluate BPC-157 in models designed to measure oxidative stress and inflammation-linked pathways, using endpoints such as lipid peroxidation markers (e.g., MDA), reactive oxygen species readouts, and nitric oxide–related biomarkers. In pharmacology-oriented studies, BPC-157 has also been explored as a mechanistic probe in models of drug-induced tissue injury, where outcomes include lesion scoring, tissue histopathology, and electrophysiology-related markers (e.g., QTc changes) in controlled preclinical settings. All findings in these domains should be interpreted as preclinical and mechanistic, dependent on model design, dosing, route, and measured endpoints.
Pathway / Mechanistic Context
Mechanistic investigations indicate that BPC-157 modulates signaling pathways involved in cellular motility and vascular organization. Experimental data suggest interaction with VEGFR2-associated signaling cascades and downstream nitric oxide–related molecular processes.
Additional in-vitro findings describe phosphorylation changes in focal adhesion signaling proteins involved in cytoskeletal anchoring and cell migration. These observations support use as a probe for cytoskeletal remodeling pathways in controlled experimental systems.
Preclinical Research Summary
Published preclinical literature documents investigations of BPC-157 across multiple experimental models. In rodent systems, the peptide has been studied in research examining vascular recruitment, gastrointestinal tissue integrity, and connective tissue remodeling under controlled laboratory conditions.
Additional investigations in avian and invertebrate models have explored peptide stability and tissue-level stress responses, contributing to broader understanding of conserved biological signaling mechanisms.
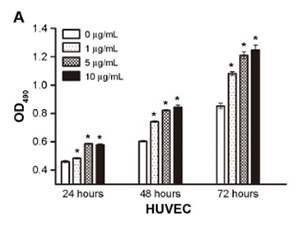 BPC-157 concentration versus vascular endothelial cell growth
BPC-157 concentration versus vascular endothelial cell growth
Source: PubMed
Form & Analytical Testing
BPC-157 is supplied as a lyophilized peptide produced via solid-phase peptide synthesis. Each lot undergoes analytical verification using high-performance liquid chromatography and mass spectrometry to confirm molecular identity and purity.
Analytical data are provided to support reproducibility and consistency in laboratory workflows.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Predrag Sikiric, lead author of “Novel Cytoprotective Mediator, Stable Gastric Pentadecapeptide BPC 157. Vascular Recruitment and Gastrointestinal Tract Healing”, and co-author of “Stable gastric pentadecapeptide BPC 157 in honeybee (Apis mellifera) therapy, to control Nosema ceranae invasions in apiary conditions,” is a Professor of Medical Department at University of Zagreb. Predrag Sikiric is listed in [9] and [16] under the referenced citations.
Predrag Sikiric is being referenced as one of the leading scientists involved in the research and development of BPC-157. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide.
Referenced Citations
- T. Huang et al., “Body protective compound-157 enhances alkali-burn wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro,” Drug Des. Devel. Ther., vol. 9, pp. 2485–2499, 2015.
- D. Drmic et al., “Counteraction of perforated cecum lesions in rats: Effects of pentadecapeptide BPC 157, L-NAME and L-arginine,” World J. Gastroenterol., vol. 24, no. 48, pp. 5462–5476, Dec. 2018.
- F. Amic et al., “Bypassing major venous occlusion and duodenal lesions in rats, and therapy with the stable gastric pentadecapeptide BPC 157, L-NAME and L-arginine,” World J. Gastroenterol., vol. 24, no. 47, pp. 5366–5378, Dec. 2018.
- A. Duzel et al., “Stable gastric pentadecapeptide BPC 157 in the treatment of colitis and ischemia and reperfusion in rats: New insights,” World J. Gastroenterol., vol. 23, no. 48, pp. 8465–8488, Dec. 2017.
- J. Vukojević et al., “Rat inferior caval vein (ICV) ligature and particular new insights with the stable gastric pentadecapeptide BPC 157,” Vascul. Pharmacol., vol. 106, pp. 54–66, 2018.
- D. Drmic et al., “Celecoxib-induced gastrointestinal, liver and brain lesions in rats, counteraction by BPC 157 or L-arginine, aggravation by L-NAME,” World J. Gastroenterol., vol. 23, no. 29, pp. 5304–5312, Aug. 2017.
- M.-J. Hsieh et al., “Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation,” J. Mol. Med. Berl. Ger., vol. 95, no. 3, pp. 323–333, 2017.
- Z. Grabarevic et al., “The influence of BPC 157 on nitric oxide agonist and antagonist induced lesions in broiler chicks,” J. Physiol. Paris, vol. 91, no. 3–5, pp. 139–149, Oct. 1997.
- P. Sikiric et al., “Novel Cytoprotective Mediator, Stable Gastric Pentadecapeptide BPC 157. Vascular Recruitment and Gastrointestinal Tract Healing,” Curr. Pharm. Des., vol. 24, no. 18, pp. 1990–2001, 2018.
- S. Seiwerth et al., “BPC 157 and Standard Angiogenic Growth Factors. Gastrointestinal Tract Healing, Lessons from Tendon, Ligament, Muscle and Bone Healing,” Curr. Pharm. Des., vol. 24, no. 18, pp. 1972–1989, 2018.
- C.-H. Chang et al., “The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration,” J. Appl. Physiol., vol. 110, no. 3, pp. 774–780, Oct. 2010.
- Y.-L. Hu et al., “FAK and paxillin dynamics at focal adhesions in the protrusions of migrating cells,” Sci. Rep., vol. 4, p. 6024, Aug. 2014.
- K. Škrlec et al., “Engineering recombinant Lactococcus lactis as a delivery vehicle for BPC-157 peptide with antioxidant activities,” Appl. Microbiol. Biotechnol., vol. 102, no. 23, pp. 10103–10117, Dec. 2018.
- D. Strinic et al., “BPC 157 counteracts QTc prolongation induced by haloperidol, fluphenazine, clozapine, olanzapine, quetiapine, sulpiride, and metoclopramide in rats,” Life Sci., vol. 186, pp. 66–79, Oct. 2017.
- N. Jelovac et al., “Pentadecapeptide BPC 157 attenuates disturbances induced by neuroleptics: the effect on catalepsy and gastric ulcers in mice and rats,” Eur. J. Pharmacol., vol. 379, no. 1, pp. 19–31, Aug. 1999.
- I. Tlak Gajger et al., “Stable gastric pentadecapeptide BPC 157 in honeybee (Apis mellifera) therapy, to control Nosema ceranae invasions in apiary conditions,” J. Vet. Pharmacol. Ther., vol. 41, no. 4, pp. 614–621, Aug. 2018.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







