GHK-Cu (2mg x 60 Capsules) (Copper Tripeptide)
$165.00
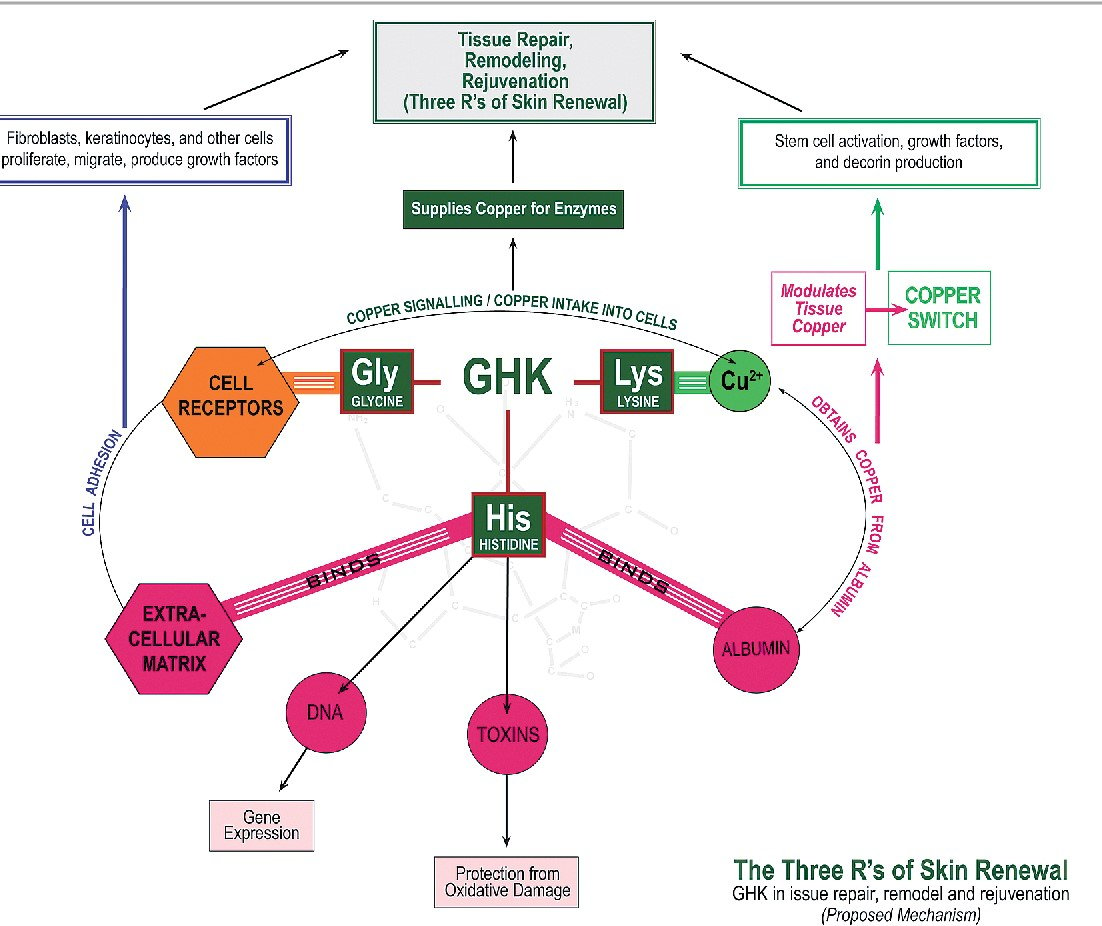
GHK-Cu is a naturally occurring copper–peptide complex composed of glycyl-histidyl-lysine bound to copper (Cu2+) and is studied for its role in cellular stress-response and gene-regulatory pathways. GHK-Cu peptide research focuses on how this small copper-chelating tripeptide influences transcriptional activity, extracellular matrix signaling, antioxidant-response networks, and inflammatory mediator expression under controlled laboratory conditions.
In vitro systems and animal models have evaluated endpoints such as collagen- and elastin-associated gene expression, cytokine signaling markers including IL-6 and TNF-related pathways, oxidative-stress indicators, proteasome-associated genes, and DNA-repair gene expression relative to controls. Major research areas include tissue remodeling assays, redox biology studies, and transcriptomic profiling across injury or stress models. These systems are central to laboratory research because copper homeostasis and extracellular matrix regulation are fundamental processes in cellular adaptation, structural maintenance, and response-to-injury mechanisms in preclinical settings.
References:
Pickart L et al., BioMed Res Int, 2015
Maquart FX et al., FEBS Lett, 1988;238(2):343–346
Siméon A et al., J Invest Dermatol, 2000;115(6):961–968
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
Overview
GHK-Cu, also known as copper glycyl-histidyl-lysine, is a small endogenous tripeptide complex in which the peptide GHK coordinates with copper ions. First identified in human plasma, it has been investigated as a copper-transporting and copper-sequestering molecule capable of influencing multiple molecular pathways in experimental systems. In research contexts, GHK-Cu is examined for its association with transcriptional regulation, extracellular matrix signaling, oxidative balance, and inflammatory-response pathways.
Copper serves as a redox-active cofactor in numerous enzymatic systems, and its coordination with peptides can influence stability, cellular distribution, and signaling interactions. Laboratory studies frequently compare GHK-Cu with peptide-alone and copper-alone controls to distinguish effects attributable to metal complexation. Much of the experimental interest centers on how copper-bound GHK may alter gene-expression programs related to tissue remodeling, protein turnover, and cellular stress adaptation under defined in vitro and preclinical conditions.
Biochemical Characteristics
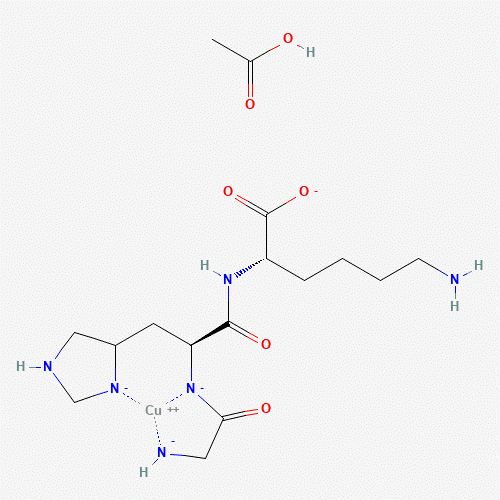
Amino Acid Sequence: Gly-His-Lys(Cu2+)
Molecular Formula: C16H28CuN6O6-2
Molecular Weight: 463.98 g/mol
PubChem CID: 156588903
CAS Number: 49557-75-7
Synonyms: copper glycyl-histidyl-lysine, lamin
 Source: PubChem
Source: PubChem
GHK coordinates Cu2+ through nitrogen- and oxygen-donor atoms contributed by the glycine N-terminus, histidine imidazole, and lysine-associated functional groups, forming a stable yet exchange-capable complex in aqueous systems. This coordination chemistry is leveraged in laboratory research to study copper partitioning between peptide ligands, proteins, and cellular compartments, as well as copper-dependent catalytic and redox processes under defined experimental conditions.
Research Applications
GHK-Cu is studied in vitro and in animal models to assess changes in molecular and cellular markers relative to appropriate control groups. Common endpoints include extracellular matrix–associated genes such as collagen and elastin targets, angiogenesis-related markers, cytokine profiles including IL-6 and TNF-related signaling, and oxidative-stress biomarkers such as reactive oxygen species–associated gene expression. Transcriptomic analyses have reported broad shifts in gene-expression patterns following exposure under controlled laboratory conditions.
Additional research examines DNA-repair–associated genes, proteasome pathway components, NF-κB–related transcriptional activity, and inflammatory mediator expression in tissue models including skin, lung, liver, and neural systems. In certain experimental injury models, investigators measure histological remodeling markers, vascularization indicators, and inflammatory cell markers compared with untreated or vehicle-treated controls. These findings are interpreted strictly within the parameters of the model system used and are confined to preclinical research settings, without implying therapeutic application or clinical use.
Pathway / Mechanistic Context
Mechanistically, GHK-Cu is studied as a copper-delivery and copper-buffering complex that can influence copper-dependent enzymes and signaling pathways. Preclinical literature describes modulation of oxidative stress pathways via changes in redox-active copper availability and downstream regulation of antioxidant defenses. GHK-Cu has also been associated with altered activity of inflammatory signaling nodes, including NF-κB–linked transcriptional programs, in cell and animal models.
Gene-expression studies have reported broad transcriptional shifts after GHK-Cu exposure in vitro, including changes in genes associated with DNA repair, proteostasis, and extracellular matrix organization. These observations are used to interrogate how copper-ligand complexes can reshape cellular stress responses and remodeling programs at the transcriptional level, including potential epigenetic contributors to metal-responsive gene regulation.
In protein-aggregation research, copper is a key variable in redox chemistry and aggregation kinetics for several amyloidogenic proteins. Laboratory studies evaluate whether copper sequestration by GHK can modify copper-catalyzed oxidative reactions and aggregation behavior under defined conditions. Such work is framed strictly as mechanistic interrogation of metal-ion contributions to protein misfolding and aggregate formation.
Source: Semantic Scholar
Preclinical Research Summary
Preclinical studies of GHK-Cu include in vitro experiments across fibroblast, endothelial, epithelial, and immune-relevant cell models, as well as in vivo animal studies in which injury-responsive endpoints are quantified. Reported findings commonly include changes in ECM-associated transcription/protein markers (e.g., collagen/elastin-related signals), modulation of inflammatory cytokine signaling (including pathways involving TNF-α and IL-6), and altered oxidative stress parameters consistent with engagement of antioxidant response pathways.
Additional animal-model literature describes changes in angiogenesis-associated signaling and remodeling markers in tissue injury paradigms. Separately, transcriptomic analyses described in the cited literature report that GHK-Cu exposure can shift expression of a substantial subset of measured genes in vitro, supporting its use as a laboratory tool for probing copper-linked transcriptional regulation and downstream pathway enrichment patterns. Research on metal-ion involvement in amyloidogenic protein chemistry has also examined GHK-Cu as a copper-sequestering variable in controlled aggregation and toxicity assay designs.
All summaries above refer only to controlled preclinical investigations and are provided to support experimental design considerations, mechanistic hypothesis generation, and pathway mapping in laboratory settings.
Form & Analytical Testing
GHK-Cu is supplied as a research-grade peptide–metal complex. Identity and composition are commonly assessed using analytical methods such as HPLC for purity profiling and mass spectrometry for molecular confirmation, with copper content/stoichiometry evaluated where applicable using techniques such as ICP-MS/ICP-OES or other validated elemental analysis approaches. UV-Vis or related spectroscopic methods may be used to characterize copper coordination features in solution under defined laboratory conditions.
Researchers should handle peptide–metal complexes using standard laboratory practices appropriate for synthetic peptides and transition-metal coordination compounds, including controls for metal contamination, chelator compatibility, and buffer composition effects on copper speciation during experimental setup.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Loren Pickart, Ph.D. has released 109 publications and is developing patents and analyzing GHK’s effects on human gene expression of 4,192 genes. In addition to GHK’s published potential uses on skin inflammation, metastatic cancer and COPD, it appears to have beneficial effects on other tissue systems such as the nervous system, gastrointestinal system, and mitochondrial system. His brief but detailed autobiography dives into the motivations and background behind his dedicating to skin, anti-aging, and life-long training.
Loren Pickart, Ph.D is being referenced as one of the leading scientists involved in the research and development of GHK-Cu. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Loren Pickart, Ph.D is listed in [3] [4] and [8] under the referenced citations.
Referenced Citations
- S. O. Canapp et al., “The effect of topical tripeptide-copper complex on healing of ischemic open wounds,” Vet. Surg. VS, vol. 32, no. 6, Art. no. 6, Dec. 2003, doi: 10.1111/j.1532-950x.2003.00515.x.
- Y. Dou, A. Lee, L. Zhu, J. Morton, and W. Ladiges, “The potential of GHK as an anti-aging peptide,” Aging Pathobiol. Ther., vol. 2, no. 1, pp. 58–61, Mar. 2020, doi: 10.31491/apt.2020.03.014.
- L. Pickart, “The human tri-peptide GHK and tissue remodeling,” J. Biomater. Sci. Polym. Ed., vol. 19, no. 8, pp. 969–988, 2008, doi: 10.1163/156856208784909435.
- L. Pickart and A. Margolina, “Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data,” Int. J. Mol. Sci., vol. 19, no. 7, p. 1987, Jul. 2018, doi: 10.3390/ijms19071987.
- S. Montes, S. Rivera-Mancia, A. Diaz-Ruiz, L. Tristan-Lopez, and C. Rios, “Copper and Copper Proteins in Parkinson’s Disease,” Oxid. Med. Cell. Longev., vol. 2014, p. e147251, Jan. 2014, doi: 10.1155/2014/147251.
- H. Kozlowski, M. Luczkowski, M. Remelli, and D. Valensin, “Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases),” Coord. Chem. Rev., vol. 256, no. 19, pp. 2129–2141, Oct. 2012, doi: 10.1016/j.ccr.2012.03.013.
- K. Rajasekhar, C. Madhu, and T. Govindaraju, “Natural Tripeptide-Based Inhibitor of Multifaceted Amyloid β Toxicity,” ACS Chem. Neurosci., vol. 7, no. 9, pp. 1300–1310, Sep. 2016, doi: 10.1021/acschemneuro.6b00175.
- L. Pickart, J. M. Vasquez-Soltero, and A. Margolina, “GHK and DNA: Resetting the Human Genome to Health,” BioMed Res. Int., vol. 2014, p. 151479, 2014, doi: 10.1155/2014/151479.
- Y. Wu, K. Cao, W. Zhang, G. Zhang, and M. Zhou, “Protective and Anti-Aging Effects of 5 Cosmeceutical Peptide Mixtures on Hydrogen Peroxide-Induced Premature Senescence in Human Skin Fibroblasts,” Skin Pharmacol. Physiol., vol. 34, no. 4, pp. 194–202, 2021, doi: 10.1159/000514496.
- X. Yang, Y. Zhang, C. Huang, L. Lu, J. Chen, and Y. Weng, “Biomimetic Hydrogel Scaffolds with Copper Peptide-Functionalized RADA16 Nanofiber Improve Wound Healing in Diabetes,” Macromol. Biosci., vol. 22, no. 8, p. e2200019, Aug. 2022, doi: 10.1002/mabi.202200019.
- X. Wang et al., “GHK-Cu-liposomes accelerate scald wound healing in mice by promoting cell proliferation and angiogenesis,” Wound Repair Regen. Off. Publ. Wound Heal. Soc. Eur. Tissue Repair Soc., vol. 25, no. 2, pp. 270–278, Apr. 2017, doi: 10.1111/wrr.12520.
- Q. Zhang, L. Yan, J. Lu, and X. Zhou, “Glycyl-L-histidyl-L-lysine-Cu2+ attenuates cigarette smoke-induced pulmonary emphysema and inflammation by reducing oxidative stress pathway,” Front. Mol. Biosci., vol. 9, p. 925700, 2022, doi: 10.3389/fmolb.2022.925700.
- J.-R. Park, H. Lee, S.-I. Kim, and S.-R. Yang, “The tri-peptide GHK-Cu complex ameliorates lipopolysaccharide-induced acute lung injury in mice,” Oncotarget, vol. 7, no. 36, pp. 58405–58417, Sep. 2016, doi: 10.18632/oncotarget.11168.
- M. Kukowska, M. Kukowska-Kaszuba, and K. Dzierzbicka, “In vitro studies of antimicrobial activity of Gly-His-Lys conjugates as potential and promising candidates for therapeutics in skin and tissue infections,” Bioorg. Med. Chem. Lett., vol. 25, no. 3, pp. 542–546, Feb. 2015, doi: 10.1016/j.bmcl.2014.12.029.
- W. Shen and T. Matsui, “Intestinal absorption of small peptides: a review,” Int. J. Food Sci. Technol., vol. 54, no. 6, pp. 1942–1948, 2019, doi: 10.1111/ijfs.14048.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.