IGF-1 LR3 (Receptor Grade) 100mcg x 10 Vials
$300.00
IGF-1 LR3 Receptor Grade is a synthetic research peptide analog of human insulin-like growth factor-1 engineered with a 13–amino acid N-terminal extension to enhance receptor affinity and reduce interaction with IGF-binding proteins. In experimental systems, IGF-1 LR3 activates the IGF-1 receptor, initiating PI3K/Akt and MAPK/ERK signaling pathways involved in cellular proliferation, differentiation, and metabolic regulation. IGF-1 LR3 Receptor Grade is utilized as a high-purity research reagent in studies examining IGF receptor kinetics, growth factor signaling, and peptide–receptor interactions under controlled laboratory conditions.
For research use only. Not for human consumption.
References:
Yakar S et al., Endocr Rev, 2001 22(6):803–817
Bach LA et al., Mol Cell Endocrinol, 2018 473:1–9
Humbel RE et al., Eur J Biochem, 1990 190(3):445–462
Overview
IGF-1 LR3 (Long Arg3 Insulin-Like Growth Factor-1) is a synthetic peptide analog engineered for laboratory investigation of insulin-like growth factor receptor (IGF-1R) signaling. Structural modification relative to native IGF-1 reduces affinity for insulin-like growth factor binding proteins (IGFBPs), resulting in altered receptor engagement kinetics and extended availability in controlled experimental systems.
This Receptor Grade material is supplied as a research reagent intended for mechanistic studies of IGF-1R-mediated signal transduction, cellular proliferation pathways, and survival signaling networks in in-vitro and preclinical in-vivo (animal) models.
Biochemical Characteristics
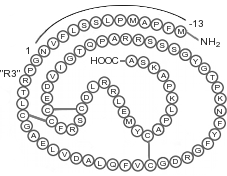
Sequence: MFPAMPLSSLFVNGPRTLCGAELVDALQFVCGDRGFYFNKPTGYGSSSRRAPQTGIVDECCFRSCDLRRLEMYCAPLKPAKSA
Molar Mass: 9,111 Da
Synonyms: Long Arg3 IGF-1 (Receptor Grade), Long R3 IGF-1
IGF-1 LR3 differs from native IGF-1 through amino-acid substitution and N-terminal extension, which collectively reduce IGFBP interaction while preserving high-affinity binding to IGF-1R. These properties make IGF-1 LR3 a useful probe for isolating receptor-specific signaling events without confounding sequestration by endogenous binding proteins in experimental matrices.
Research Applications
- IGF-1 receptor binding and activation assays
- Signal transduction studies involving PI3K/AKT and MAPK/ERK pathways
- Comparative analyses of IGFBP-dependent versus IGFBP-resistant IGF analogs
- Cell proliferation, differentiation, and apoptosis modeling in cultured cell systems
- Receptor kinetics and downstream phosphorylation mapping
- Preclinical in-vivo (animal) studies examining IGF-axis biology
Receptor Grade IGF-1 LR3 is commonly selected for experiments requiring robust and sustained IGF-1R engagement under tightly controlled laboratory conditions.
Pathway / Mechanistic Context
IGF-1 LR3 primarily interacts with the insulin-like growth factor-1 receptor (IGF-1R), a transmembrane tyrosine kinase receptor. Ligand binding induces receptor autophosphorylation, initiating downstream signaling cascades including:
- PI3K → AKT signaling associated with cellular survival and metabolic regulation
- RAS → RAF → MEK → ERK signaling involved in cell cycle progression and differentiation
Due to its reduced affinity for IGFBPs, IGF-1 LR3 demonstrates prolonged receptor availability in experimental systems, enabling extended interrogation of receptor-proximal and downstream signaling events. Limited cross-interaction with the insulin receptor has also been reported in biochemical assays, providing additional context for comparative receptor specificity studies.
Preclinical Research Summary
Preclinical investigations utilizing IGF-1 LR3 focus on elucidating IGF-axis biology rather than organism-level outcomes. In cell-based and animal models, IGF-1 LR3 has been employed to:
- Characterize receptor binding affinity and internalization dynamics
- Quantify downstream phosphorylation profiles of IGF-1R substrates
- Model regulatory mechanisms governing cell survival and programmed cell death
- Examine differentiation pathways in lineage-specific progenitor cells
All findings reported in the literature are derived from controlled in-vitro or animal-based experimental systems and are interpreted strictly within a mechanistic research framework.
Form & Analytical Testing
This product is supplied as a lyophilized peptide for laboratory research use. Analytical characterization typically includes identity confirmation and purity assessment using orthogonal methods such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS).
Researchers should employ standard peptide handling practices, including controlled reconstitution, appropriate buffer selection, and minimization of adsorption or degradation, to ensure experimental consistency.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Anastasios Philippou, Ph.D. focused on Experimental Physiology at the National & Kapodistrian University of Athens Medical School. He is now a National Center Manager and Assistant Professor, however his extensive studying and documented research pertaining to the effects of muscle regeneration, the role of IGF-1 in skeletal muscle physiology, the expression of IGF-1 isoforms after exercise induced muscle damage in humans, characterization of the MGF E peptide actions in vitro, and epigenetic regulation on gene expression induced by physical exercise are most impressive.
Dr. Anastasios Philippou, Ph.D. is being referenced as one of the leading scientists involved in the research and development of IGF1-LR3. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Anastasios Philippou, Ph.D. is listed in [7] and [8] under the referenced citations.
Referenced Citations
- “Adipose Tissue-Derived Stem Cell Secreted IGF-1 Protects Myoblasts from the Negative Effect of Myostatin.” [Online]. Available: https://www.hindawi.com/journals/bmri/2014/129048/. [Accessed: 16-May-2019].
- N. Li, Q. Yang, R. G. Walker, T. B. Thompson, M. Du, and B. D. Rodgers, “Myostatin Attenuation In Vivo Reduces Adiposity, but Activates Adipogenesis,” Endocrinology, vol. 157, no. 1, pp. 282–291, Jan. 2016.
- E. Corpas, S. M. Harman, and M. R. Blackman, “Human growth hormone and human aging,” Endocr. Rev., vol. 14, no. 1, pp. 20–39, Feb. 1993.
- W. E. Sonntag, A. Csiszar, R. deCabo, L. Ferrucci, and Z. Ungvari, “Diverse roles of growth hormone and insulin-like growth factor-1 in mammalian aging: progress and controversies,” J. Gerontol. A. Biol. Sci. Med. Sci., vol. 67, no. 6, pp. 587–598, Jun. 2012.
- “IGF-I/IGFBP system: metabolism outline and physical exercise. – PubMed – NCBI.” [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/22714057. [Accessed: 16-May-2019].
- B. Y. Hanaoka, C. A. Peterson, C. Horbinski, and L. J. Crofford, “Implications of glucocorticoid therapy in idiopathic inflammatory myopathies,” Nat. Rev. Rheumatol., vol. 8, no. 8, pp. 448–457, Aug. 2012.
- A Philippou, A Halapas, M Maridaki, M Koutsilieris – J Musculoskelet Neuronal Interact, 2007 [Semantic Scholar]
- A Philippou, E Papageorgiou, G Bogdanis, A Halapas… – In vivo, 2009 [Iiar Journals]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







