Melanotan 1 (MT1) 10mg
$55.00
Melanotan I is a synthetic linear α-melanocyte-stimulating hormone (α-MSH) analog (Ac-Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH₂) designed to study melanocortin receptor activation, particularly MC1R. It activates cAMP-dependent signaling cascades involved in pigmentation and photoprotective pathways in vitro. Melanotan I is widely used in receptor pharmacology and cell signaling research investigating peptide–GPCR interactions and transcriptional regulation.
For research use only. Not for human consumption.
References:
Abdel-Malek ZA et al., FASEB J, 1995 9(12):1175–1180
Mountjoy KG et al., Endocr Rev, 1994 15(2):135–155
Hadley ME et al., Peptides, 1996 17(4):593–596
Overview
Melanotan 1 is a synthetic analogue of alpha-melanocyte stimulating hormone (α-MSH) used as a laboratory research reagent to investigate melanocortin receptor biology. In mechanistic systems, melanotan 1 is used as an agonist probe for melanocortin receptors (MC1R, MC3R, MC4R, and MC5R) to evaluate receptor activation, downstream second-messenger signaling (e.g., cAMP/PKA), and associated transcriptional responses in controlled in-vitro assays and in-vivo animal models.
Due to its structural similarity to α-MSH, melanotan 1 is frequently incorporated into comparative pharmacology experiments to examine ligand–receptor selectivity, potency/efficacy relationships, and pathway outputs across melanocortin receptor subtypes under standardized assay conditions.
Biochemical Characteristics
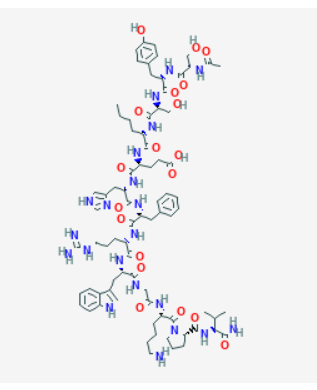 Source: PubChem
Source: PubChem
Sequence: Ser-Tyr-Ser-Nle-Glu-His-D-Phe-Arg-Trp-Gly-Lys-Pro-Val
Molecular Formula: C78H111N21O19
Molecular Weight: 1646.874 g/mol
PubChem CID: 16154396
CAS Number: 75921-69-6
Synonyms: Afamelanotide, CUV1657, MT-1
Melanotan 1 is a peptide engineered to retain key α-MSH pharmacophore features while incorporating substitutions (e.g., Nle at position 4 and D-Phe at position 7) commonly used in peptide chemistry to modulate stability and receptor interaction profiles in experimental systems. These modifications are routinely evaluated using receptor-binding and signaling assays across melanocortin receptor subtypes.
Research Applications
1. Melanocortin Receptor Pharmacology & Second-Messenger Readouts
Melanotan 1 is used in cell-based receptor assays to quantify melanocortin receptor activation using second-messenger endpoints such as cAMP accumulation, PKA-linked reporter activity, and immediate early gene expression. Assays may be performed in recombinant expression systems or in cells endogenously expressing melanocortin receptors, with subtype attribution supported by antagonist controls and receptor-selective tool compounds.
2. Pigment-Pathway Signaling in Melanocyte-Associated Model Systems
In melanocyte-focused experimental models, melanotan 1 is used to examine MC1R-linked signaling networks and transcriptional regulation of melanogenesis-associated enzyme pathways. Common readouts include cAMP dynamics, CREB-associated transcriptional activity, melanogenic gene expression panels, and melanosome biology endpoints measured in vitro.
3. Neurobiology & Neuroinflammation-Linked Pathway Mapping (Preclinical)
Melanocortin agonists are used in rodent models to interrogate CNS-relevant melanocortin signaling, including pathway panels associated with synaptic signaling, neurotrophin-linked transcriptional programs (e.g., BDNF-associated pathways), and inflammation-linked molecular endpoints. Study designs commonly incorporate receptor subtype modulation (often MC4R-focused paradigms) to support mechanistic attribution.
4. Cardiovascular and Autonomic Signaling Endpoints (Preclinical)
In animal studies, melanocortin receptor agonists have been evaluated in experimental designs that quantify vascular tone, autonomic regulation, and hemodynamic parameters as mechanistic outputs. These models are used to examine receptor subtype involvement and downstream signaling mediators in controlled preclinical settings.
5. Metabolic Pathway Interrogation (Preclinical)
Because melanocortin receptors participate in energy homeostasis circuits, melanotan 1 may be used as a receptor agonist probe in preclinical models evaluating downstream metabolic signaling networks. Experimental endpoints may include receptor-dependent second messenger signaling, gene expression signatures, and tissue-specific pathway activation associated with lipid handling and substrate utilization.
Pathway / Mechanistic Context
Melanotan 1 engages the melanocortin receptor family, a GPCR class commonly coupled to Gs-associated signaling and cAMP generation. In receptor characterization studies, melanotan 1 is used to map cAMP/PKA pathway activation, CREB-linked transcriptional effects, and receptor desensitization/internalization behavior using time-course assays. Depending on receptor subtype and cellular context, melanocortin receptor activation can also be evaluated for cross-talk with MAPK/ERK and other intracellular signaling cascades using phosphorylation-state panels and transcriptional profiling.
Because melanotan 1 can activate multiple melanocortin receptor subtypes, mechanistic workflows commonly incorporate subtype-selective antagonists/agonists and receptor-expression controls to deconvolute receptor contributions to measured biochemical endpoints (e.g., cAMP accumulation, ERK phosphorylation, and transcriptomic signatures) in cell-based assays and animal models.
Preclinical Research Summary
Preclinical research evaluating melanocortin agonists (including α-MSH analogs) includes rodent studies of blood pressure and vascular signaling endpoints, neuroinflammation-linked models, and CNS pathway mapping designs that quantify astrocyte/neuron signaling markers, neurotrophin-associated transcriptional readouts, and ex-vivo molecular correlates.[5], [12], [9]
Additional animal studies have assessed melanocortin pathway modulation in models of ischemic injury and cardiac arrest, using molecular and functional endpoints appropriate to the preclinical design.[10], [11] All references are provided strictly as mechanistic background for laboratory research contexts.
Form & Analytical Testing
Analytical characterization in RUO workflows commonly includes identity confirmation and purity profiling using chromatographic and mass-based methods (e.g., HPLC and MS). When provided, lot-specific documentation is used for laboratory quality review of identity, purity, and consistency within the experimental context.
Referenced Citations
- “α-MSH analogue attenuates blood pressure elevation in DOCA-salt hypertensive mice. – PubMed – NCBI.”
- “Melanocortins protect against brain damage and counteract cognitive decline in a transgenic mouse model of moderate Alzheimer׳s disease. – PubMed – NCBI.”
- “NDP-α-MSH induces intense neurogenesis and cognitive recovery in Alzheimer transgenic mice through activation of melanocortin MC4 receptors. – PubMed – NCBI.”
- “Melanocortins protect against progression of Alzheimer’s disease in triple-transgenic mice by targeting multiple pathophysiological pathways. – PubMed – NCBI.”
- “Melanocortin 4 receptor activates ERK-cFos pathway to increase brain-derived neurotrophic factor expression in rat astrocytes and hypothalamus. – PubMed – NCBI.”
- “Functional recovery after delayed treatment of ischemic stroke with melanocortins is associated with overexpression of the activity-dependent gene … – PubMed – NCBI.”
- “Protective effects of the melanocortin analog NDP-α-MSH in rats undergoing cardiac arrest. – PubMed – NCBI.”
- “Melanocortin-1 receptor activation is neuroprotective in mouse models of neuroinflammatory disease. – PubMed – NCBI.”
- “Immunosuppressive activity of a novel peptide analog of α-melanocyte stimulating hormone (α-MSH) in experimental autoimmune uveitis. – PubMed – NCBI.”
- “Peripheral effect of alpha-melanocyte-stimulating hormone on fatty acid oxidation in skeletal muscle. – PubMed – NCBI.”
- “Characterization of murine melanocortin receptors mediating adipocyte lipolysis and examination of signalling pathways involved. – PubMed – NCBI.”
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







