Melanotan 2 (MT2) 10mg
$50.00
Melanotan 2 is a synthetic cyclic heptapeptide (Ac-Nle-c[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂) that functions as a potent nonselective melanocortin receptor agonist. It is employed in receptor binding and signal transduction studies exploring MC1R–MC5R-mediated cAMP activation and GPCR desensitization mechanisms. MT-II provides a research model for peptide–receptor pharmacology and neuroendocrine signaling dynamics.
For research use only. Not for human consumption.
References:
Hadley ME et al., Peptides, 1996 17(4):593–596
Mountjoy KG et al., Endocr Rev, 1994 15(2):135–155
Hruby VJ et al., Peptides, 2000 21(3):423–438
Overview
Melanotan 2 (MT-2) is a synthetic analogue of human alpha-melanocyte-stimulating hormone (α-MSH) used as a laboratory research reagent to investigate melanocortin receptor biology. In controlled experimental settings, MT-2 is used to evaluate receptor activation, downstream second-messenger signaling, and pathway-linked transcriptional responses across melanocortin-associated systems.
MT-2 is frequently discussed in research literature in the context of melanocortin-driven pathways relevant to pigmentation biology and CNS-linked behavioral signaling. All information provided here is presented strictly for informational and educational research-context purposes.
Biochemical Characteristics
Peptide Sequence: Nle-Asp(1)-His-D-Phe-Arg-Trp-Lys(1)
Molecular Formula: C50H69N15O9
Molecular Weight: 1024.198 g/mol
PubChem CID: 92432
CAS Number: 121062-08-6
 Source: PubChem
Source: PubChem
MT-2 is typically evaluated as a defined-sequence melanocortin agonist input in receptor binding, signaling, and time-course pathway assays.
Research Applications
In research settings, MT-2 may be used as an experimental input for:
- Receptor pharmacology: comparing potency/efficacy across melanocortin receptor subtypes in cell-based assays.
- Second-messenger signaling: quantifying pathway outputs such as cAMP accumulation and downstream kinase activation (e.g., ERK) under controlled conditions.
- Behavioral neuroscience models: probing melanocortin-linked circuitry in preclinical paradigms studying feeding behavior, reward/impulse-related behavior, and other neurobehavioral endpoints.
- Pigmentation biology: investigating melanocyte-associated signaling and transcriptional programs downstream of melanocortin receptor activation.
- Systems biology / mapping studies: evaluating tissue- or region-specific receptor expression and pathway signatures using agonist-stimulation frameworks.
These applications are presented as examples of laboratory research use and are not medical or clinical claims.
Pathway / Mechanistic Context
MT-2 is discussed as engaging the melanocortin receptor family (MC1R–MC5R), a group of GPCRs that in many experimental systems couple to Gs signaling and drive cAMP generation. In mechanistic workflows, MT-2 is used to map receptor-driven pathway readouts such as cAMP/PKA-linked signaling, ERK phosphorylation, immediate-early gene expression, and downstream transcriptional programs.
Because melanocortin receptors have distinct expression patterns across tissues, MT-2 is often incorporated alongside subtype-selective antagonists, genetic models, or region-specific interventions to support receptor attribution of observed endpoints.
- MC1R: commonly studied in melanocyte and pigmentation signaling models.
- MC3R: investigated in energy-balance and metabolic signaling contexts.
- MC4R: frequently used as a mechanistic anchor in feeding behavior and neurobehavioral pathway studies.
- MC5R: explored in peripheral tissues, supporting tissue-distribution and signaling studies.
Preclinical Research Summary
1. Neurodevelopmental Behavior Models (MIA / ASD-related paradigms)
In a maternal immune activation (MIA) mouse model, MT-2 administration was reported alongside changes in measured autism-like behavioral endpoints and oxytocin-related signaling readouts, supporting continued investigation of melanocortin-linked circuitry in social-behavior paradigms.[1]
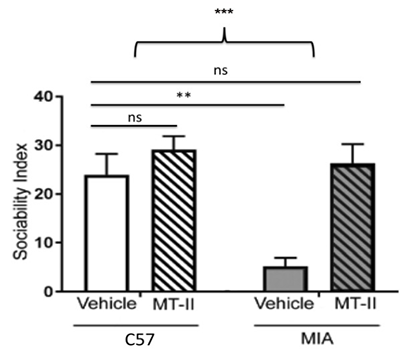 Impact of MT-2 on sociability in rats with ASD (MIA) showing that MT-2 returns sociability ratings to near the baseline of control animals (C57).
Impact of MT-2 on sociability in rats with ASD (MIA) showing that MT-2 returns sociability ratings to near the baseline of control animals (C57).
Source: PubMed
2. Feeding Behavior, Macronutrient Preference, and Satiety Pathways
Human and animal research has examined melanocortin signaling (including MC4R-linked paradigms) for relationships to dietary intake patterns and macronutrient preference.[2] Additional literature discusses leptin–melanocortin network interactions relevant to feeding and energy expenditure regulation.[3], [4] Related work has also examined TRH gene regulation within these broader metabolic circuits.[5]
3. Metabolic Regulation & Glucose-Handling Literature
Metabolic research discusses glucagon as a factor in diabetes pathophysiology and explores CNS-linked signaling contributions to peripheral glucose handling.[6] In hypothalamic-nuclei injection models, melanocortin receptor agonism has been evaluated for effects on glucose uptake endpoints in peripheral tissues, supporting ongoing interest in central regulation of metabolic function.[7]
4. Reward / Impulse-Related Behavioral Research
Rodent studies have reported that melanocortin signaling in regions such as the amygdala may influence alcohol intake behaviors, with MT-2 used as a receptor-agonist probe in these paradigms.[8] Additional work has evaluated combined-intervention designs where MT-2 is assessed alongside naltrexone in binge-like ethanol intake models in mice.[9]
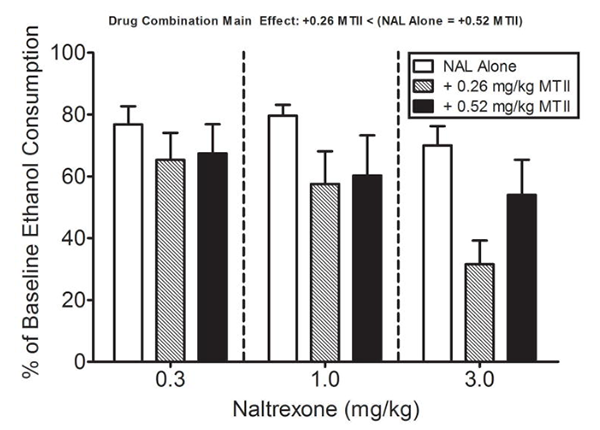 Percent of baseline alcohol consumption in mice treated with naltrexone or naltrexone and MT-2.
Percent of baseline alcohol consumption in mice treated with naltrexone or naltrexone and MT-2.
Source: PubMed
5. Sexual Behavior Signaling (Clinical and Preclinical Literature)
Clinical research has evaluated MT-2 in the context of psychogenic erectile dysfunction, including a placebo-controlled crossover study reporting effects on erection initiation under the study’s measured conditions.[10]
Note: The studies summarized above describe mechanistic or experimental endpoints in laboratory and preclinical systems and are not intended as medical claims.
Form & Analytical Testing
For laboratory workflows, peptide identity and quality are commonly evaluated using standard analytical approaches (e.g., chromatographic purity profiling and mass-based identity confirmation) to support controlled experimental use. Analytical outputs are interpreted within the purchaser’s quality framework and study requirements.
This section is provided for general informational context regarding common laboratory quality practices and does not represent a medical, diagnostic, or clinical application.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Wessells is a University of Washington faculty member in urology and has published research related to urogenital trauma and erectile dysfunction. He has been associated with research on pro-erectile melanocortin agonists studied for erectile dysfunction.
Dr. Wessells is referenced here to acknowledge scientific contributions in this research area. This reference does not imply endorsement or advocacy of purchase, sale, or use of this product. No affiliation or relationship is implied between the seller and this scientist. Dr. Wessells is referenced within the citations below.
Referenced Citations
- E. Minakova et al., “Melanotan-II reverses autistic features in a maternal immune activation mouse model of autism,” PLoS ONE, vol. 14, no. 1, Jan. 2019.
- A. van der Klaauw et al., “Role of melanocortin signalling in the preference for dietary macronutrients in human beings,” Lancet, vol. 385 Suppl 1, p. S12, Feb. 2015.
- H. Shimizu, K. Inoue, and M. Mori, “The leptin-dependent and -independent melanocortin signaling system: regulation of feeding and energy expenditure,” J. Endocrinol., vol. 193, no. 1, pp. 1–9, Apr. 2007.
- C. Bjørbaek and A. N. Hollenberg, “Leptin and melanocortin signaling in the hypothalamus,” Vitam. Horm., vol. 65, pp. 281–311, 2002.
- F. Guo, K. Bakal, Y. Minokoshi, and A. N. Hollenberg, “Leptin Signaling Targets the Thyrotropin-Releasing Hormone Gene Promoter in Vivo,” Endocrinology, vol. 145, no. 5, pp. 2221–2227, May 2004.
- Y. H. Lee, M.-Y. Wang, X.-X. Yu, and R. H. Unger, “Glucagon is the key factor in the development of diabetes,” Diabetologia, vol. 59, no. 7, pp. 1372–1375, 2016.
- C. Toda et al., “Distinct effects of leptin and a melanocortin receptor agonist injected into medial hypothalamic nuclei on glucose uptake in peripheral tissues,” Diabetes, vol. 58, no. 12, pp. 2757–2765, Dec. 2009.
- D. A. York, S. Boghossian, and M. Park-York, “Melanocortin activity in the amygdala influences alcohol intake,” Pharmacol. Biochem. Behav., vol. 98, no. 1, pp. 112–119, Mar. 2011.
- M. Navarro, F. Carvajal, J. M. Lerma-Cabrera, I. Cubero, M. J. Picker, and T. E. Thiele, “Evidence that Melanocortin Receptor Agonist Melanotan-II Synergistically Augments the Ability of Naltrexone to Blunt Binge-Like Ethanol Intake in Male C57BL/6J Mice,” Alcohol. Clin. Exp. Res., vol. 39, no. 8, pp. 1425–1433, Aug. 2015.
- “Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: double-blind, placebo controlled crossover study.” PubMed (PMID: 9679884).
- H. Wessells et al., “MT-II Induces Penile Erection via Brain and Spinal Mechanisms,” Annals of the New York Academy of Sciences, 994: 90–95, 2003.
- Wessells, H. (1998). Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: Doubleblind placebo controlled crossover study. Nature.com PDF.
- M. T. Islam et al., “Vasopressin neurons in the paraventricular hypothalamus promote wakefulness via lateral hypothalamic orexin neurons,” Curr. Biol., Jul. 2022.
- J. K. Y. Lau et al., “Melanocortin receptor activation alleviates amyloid pathology and glial reactivity in an Alzheimer’s disease transgenic mouse model,” Sci. Rep., Feb. 2021.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







