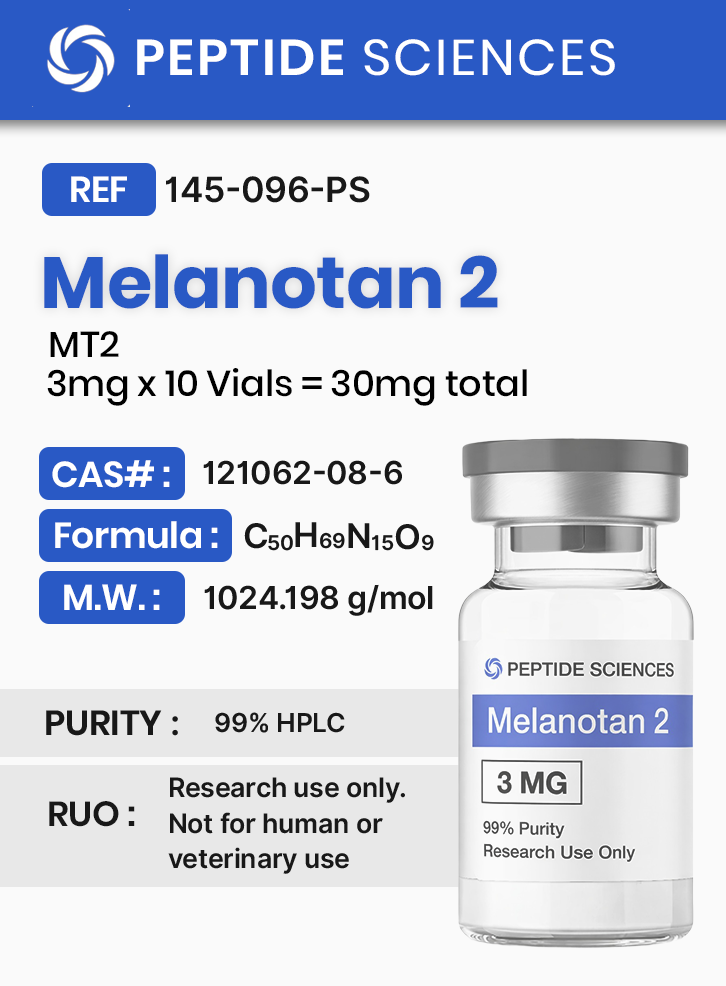
Melanotan 2 (MT2) (3mg x 10 Vials = 30mg)
$140.00
Melanotan-2 (MT-2) is a synthetic cyclic heptapeptide (Ac-Nle-c[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂) that acts as a potent nonselective melanocortin receptor agonist. It is used in receptor binding and signal transduction research to investigate MC1R–MC5R pathways related to pigmentation, energy balance, and neuroendocrine signaling. MT-II provides a model for studying GPCR-mediated cAMP activation and receptor desensitization.
For research use only. Not for human consumption.
References:
Fan W et al., Endocrinology, 1997;138(6):2483–2489
Mountjoy KG et al., Endocr Rev, 1994;15(2):135–155
Hruby VJ et al., Peptides, 2000;21(3):423–438
Overview
Melanotan 2 (MT-2) is a synthetic cyclic heptapeptide structurally derived from the endogenous melanocortin peptide α-melanocyte-stimulating hormone (α-MSH). It is employed in laboratory research as a non-selective agonist of melanocortin receptors and serves as a molecular tool for investigating melanocortin signaling pathways in cellular and in-vivo experimental systems.
Preclinical research utilizes MT-2 to interrogate receptor-mediated signal transduction, neuropeptide-driven transcriptional programs, and downstream second-messenger cascades associated with melanocortin receptor activation in controlled experimental models.
Biochemical Characteristics
Peptide Sequence: Nle-Asp-His-D-Phe-Arg-Trp-Lys
Molecular Formula: C50H69N15O9
Molecular Weight: 1024.198 g/mol
PubChem CID: 92432
CAS Number: 121062-08-6
Research Applications
MT-2 is used in research environments to study melanocortin receptor activation across multiple tissue types and neural circuits. Experimental applications include investigation of receptor-specific signaling bias, ligand–receptor affinity profiles, and downstream transcriptional responses mediated by melanocortin receptor subtypes.
Additional research contexts include analysis of neuroendocrine signaling networks, hypothalamic peptide integration, and receptor-driven modulation of intracellular signaling pathways in rodent and cellular model systems.
Pathway / Mechanistic Context
Melanotan 2 functions as an agonist at multiple melanocortin receptor subtypes, including MC1R, MC3R, MC4R, and MC5R. These G-protein-coupled receptors initiate intracellular signaling cascades primarily through cyclic AMP (cAMP) generation and downstream protein kinase activation.
Mechanistic studies examine MT-2-induced receptor activation to characterize melanocortin-dependent modulation of transcriptional regulators, neuronal firing patterns, neuropeptide release dynamics, and metabolic signaling integration in defined experimental systems.
Preclinical Research Summary
Preclinical investigations utilizing MT-2 have explored melanocortin receptor signaling in rodent behavioral paradigms, neuroendocrine circuits, and metabolic signaling networks. These studies are used to map receptor distribution, signal propagation, and receptor-specific pathway engagement at molecular and cellular resolution.
Additional experimental literature evaluates MT-2-associated modulation of hypothalamic signaling nodes, limbic system activity, and central peptide integration using in-vivo animal models and in-vitro neuronal culture systems. Findings are interpreted within a mechanistic framework without translational inference.
Form & Analytical Testing
Melanotan 2 is supplied as a synthetic research peptide for controlled laboratory workflows. Peptide identity and composition may be verified using analytical techniques such as mass spectrometry and high-performance liquid chromatography in accordance with standard peptide characterization practices.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Wessells is a UW professor, chair of the Department of Urology, and has served on several national and international professional and government committees, including the WHO International Consultations on Erectile and Sexual Dysfunction, an NIDDK working group on urological complications of diabetes, and a NIH symposium on diabetes. He is a surgeon, researcher and expert on urogenital trauma and erectile dysfunction. His clinical interests include reconstructive surgery of the genitourinary tract, acute injury management and complex surgery for male sexual dysfunction. His research interests are in urogenital trauma epidemiology and management; the physiology and pathophysiology of erectile dysfunction; reconstructive surgery; crash injury mechanics; and urological complications of diabetes. A proerectile melanocortin agonist developed by Dr. Wessells and his collaborators at the University of Arizona is in clinical trials for the treatment of erectile dysfunction.
Dr. Wessells is being referenced as one of the leading scientists involved in the research and development of Melanotan 2. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Wessells is listed in [11] and [12] under the referenced citations.
Referenced Citations
- E. Minakova et al., “Melanotan-II reverses autistic features in a maternal immune activation mouse model of autism,” PLoS ONE, vol. 14, no. 1, Jan. 2019. [PubMed]
- A. van der Klaauw et al., “Role of melanocortin signaling in the preference for dietary macronutrients in human beings,” Lancet Lond. Engl., vol. 385 Suppl 1, p. S12, Feb. 2015. [PubMed]
- H. Shimizu, K. Inoue, and M. Mori, “The leptin-dependent and -independent melanocortin signaling system: regulation of feeding and energy expenditure,” J. Endocrinol., vol. 193, no. 1, pp. 1–9, Apr. 2007. [Research Gate]
- C. Bjørbaek and A. N. Hollenberg, “Leptin and melanocortin signaling in the hypothalamus,” Vitam. Horm., vol. 65, pp. 281–311, 2002. [PubMed]
- F. Guo, K. Bakal, Y. Minokoshi, and A. N. Hollenberg, “Leptin Signaling Targets the Thyrotropin-Releasing Hormone Gene Promoter in Vivo,” Endocrinology, vol. 145, no. 5, pp. 2221–2227, May 2004. [PubMed]
- Y. H. Lee, M.-Y. Wang, X.-X. Yu, and R. H. Unger, “Glucagon is the key factor in the development of diabetes,” Diabetologia, vol. 59, no. 7, pp. 1372–1375, 2016. [PubMed]
- C. Toda et al., “Distinct effects of leptin and a melanocortin receptor agonist injected into medial hypothalamic nuclei on glucose uptake in peripheral tissues,” Diabetes, vol. 58, no. 12, pp. 2757–2765, Dec. 2009. [PubMed]
- D. A. York, S. Boghossian, and M. Park-York, “Melanocortin activity in the amygdala influences alcohol intake,” Pharmacol. Biochem. Behav., vol. 98, no. 1, pp. 112–119, Mar. 2011. [PubMed]
- M. Navarro, F. Carvajal, J. M. Lerma-Cabrera, I. Cubero, M. J. Picker, and T. E. Thiele, “Evidence that Melanocortin Receptor Agonist Melanotan-II Synergistically Augments the Ability of Naltrexone to Blunt Binge-Like Ethanol Intake in Male C57BL/6J Mice,” Alcohol. Clin. Exp. Res., vol. 39, no. 8, pp. 1425–1433, Aug. 2015. [PubMed]
- “Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: double-blind, placebo controlled crossover study. – PubMed – NCBI.” [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/9679884. [Accessed: 15-May-2019].
- WESSELLS, H. , HRUBY, V. J., HACKETT, J. , HAN, G. , BALSE‐SRINIVASAN, P. and VANDERAH, T. W. (2003), MT‐II Induces Penile Erection via Brain and Spinal Mechanisms. Annals of the New York Academy of Sciences, 994: 90-95. doi:10.1111/j.1749-6632.2003.tb03166.x
- Wessells, H. (1998). Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: Doubleblind placebo controlled crossover study. Nature.com. Available at: https://www.nature.com/articles/3900371.pdf
- M. T. Islam et al., “Vasopressin neurons in the paraventricular hypothalamus promote wakefulness via lateral hypothalamic orexin neurons,” Curr. Biol. CB, pp. S0960-9822(22)01121–6, Jul. 2022, doi: 10.1016/j.cub.2022.07.020. [PubMed]
- J. K. Y. Lau et al., “Melanocortin receptor activation alleviates amyloid pathology and glial reactivity in an Alzheimer’s disease transgenic mouse model,” Sci. Rep., vol. 11, no. 1, p. 4359, Feb. 2021, doi: 10.1038/s41598-021-83932-4. [PubMed]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.