NMN 500mg (60 Capsules)
$65.00
NMN is a research compound functioning as a direct precursor to nicotinamide adenine dinucleotide (NAD⁺), a central cofactor in cellular redox metabolism. Preclinical research demonstrates its role in modulating sirtuin activity, mitochondrial biogenesis, and oxidative stress response through the NAD⁺ salvage pathway. NMN is utilized in studies investigating energy metabolism, aging-related signaling, and nicotinamide-dependent enzymatic regulation under controlled laboratory conditions.
References:
Yoshino J et al., Cell Metab, 2018 27(3):529–547
Mills KF et al., Cell Metab, 2016 24(5):795–806
Imai SI et al., Nat Rev Mol Cell Biol, 2014 15(3):223–240
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
Overview
Nicotinamide mononucleotide (NMN) is a phosphorylated nicotinamide riboside derivative and a central intermediate in nicotinamide adenine dinucleotide (NAD⁺) biosynthesis. In laboratory research, NMN is used as a biochemical tool for studying NAD⁺ salvage dynamics, redox cofactor availability, and NAD⁺-dependent enzymatic pathways in cellular and animal model systems.
Preclinical investigations evaluate NMN uptake, intracellular conversion, and downstream effects on NAD⁺-consuming enzymes using in-vitro, ex-vivo, and in-vivo animal models. All discussion herein is limited to mechanistic and pathway-level research contexts and does not extend to clinical or applied use.
Biochemical Characteristics

Chemical Formula: C11H15N2O8P
Molecular Weight: 334.22 g/mol
PubChem CID: 14180
CAS No: 1094-61-7
Synonyms: Nicotinamide-1-ium-1-β-D-ribofuranoside 5′-phosphate
Research Applications
Biochemical Role of NMN
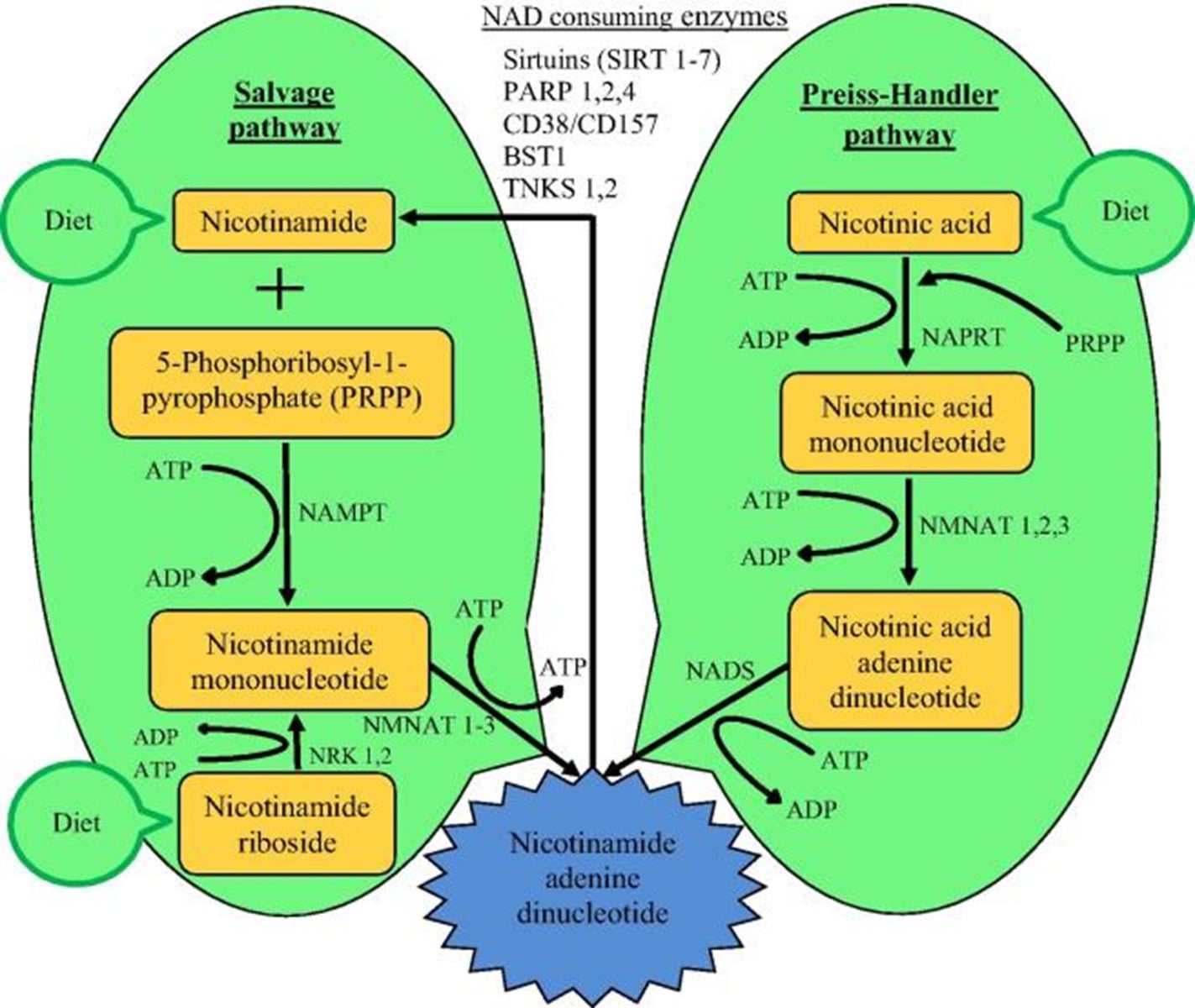
NMN functions as a key intermediate in NAD⁺ biosynthesis via the nicotinamide salvage and Preiss–Handler pathways. In experimental systems, NMN is incorporated into NAD⁺ pools through enzymatic conversion mediated by nicotinamide phosphoribosyltransferase (NAMPT) and NMN adenylyltransferases (NMNATs). These pathways are routinely studied to quantify NAD⁺ turnover, metabolic flux, and cofactor availability.
- De novo synthesis from tryptophan
- Nicotinamide salvage pathway
- Preiss–Handler pathway
Within these pathways, NMN serves as a measurable node linking precursor availability to NAD⁺ production, enabling controlled interrogation of NAD⁺-dependent cellular processes.
NAD⁺-Dependent Enzyme Systems
In preclinical models, NMN availability is evaluated in relation to NAD⁺-dependent enzyme families including sirtuins and poly(ADP-ribose) polymerases (PARPs). These enzymes are examined using biochemical assays to assess post-translational modification activity, chromatin-associated signaling, and DNA damage response pathways.
PARP activity is commonly quantified through ADP-ribosylation assays, while sirtuin-associated pathways are evaluated using deacetylation markers and downstream transcriptional readouts. NMN is used in experimental designs to modulate intracellular NAD⁺ availability and characterize enzyme-specific pathway responses.
Pathway / Mechanistic Context
NAD⁺ functions as a central redox cofactor in mitochondrial oxidative metabolism. In animal and cellular studies, NMN supplementation is used to examine mitochondrial respiration parameters, redox balance, and oxidative phosphorylation efficiency. These measurements are conducted using respirometry, enzymatic flux analysis, and mitochondrial protein expression profiling.
Preclinical Research Summary
Preclinical studies in rodents have identified transporter-mediated uptake mechanisms for NMN in intestinal and tissue-specific models. These systems are used to study NMN distribution kinetics, intracellular conversion rates, and tissue-specific NAD⁺ pool dynamics using labeled tracers and mass spectrometry–based quantification.
Research Scope Summary
Across published in-vitro and animal studies, NMN is utilized as a research reagent to investigate NAD⁺ metabolism, enzymatic cofactor utilization, mitochondrial redox balance, and genome maintenance signaling. Observed effects are interpreted exclusively through molecular, biochemical, and pathway-level endpoints without extrapolation beyond controlled experimental systems.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Gláucia Maria Pastore graduated in Biological Sciences from the Pontifical Catholic University of Campinas and earned a Master’s and PhD in Food Science from Unicamp, where she also studied Enzyme Technology in Japan and completed postdoctoral work at Ohio State University. Since 2006, she has taught Food Biochemistry at Unicamp, overseeing the Food Science Program and teaching both undergraduate and graduate courses. Her research includes 134 articles, 239 conference proceedings, six patents, and extensive leadership in food biochemistry research.
Dr. Gláucia Maria Pastore is referenced solely to acknowledge contributions to the scientific literature related to NMN and NAD⁺ metabolism. No endorsement, affiliation, or implied relationship with any commercial product is stated or suggested.
Referenced Citations
- A. Grozio et al., “Slc12a8 is a nicotinamide mononucleotide transporter,” Nat Metab, vol. 1, no. 1, pp. 47–57, Jan. 2019, doi: 10.1038/s42255-018-0009-4.
- R. Spector and C. E. Johanson, “Vitamin transport and homeostasis in mammalian brain: focus on Vitamins B and E,” J Neurochem, vol. 103, no. 2, pp. 425–438, Oct. 2007, doi: 10.1111/j.1471-4159.2007.04773.x.
- H. Nadeeshani, J. Li, T. Ying, B. Zhang, and J. Lu, “Nicotinamide mononucleotide (NMN) as an anti-aging health product – Promises and safety concerns,” J Adv Res, vol. 37, pp. 267–278, Mar. 2022, doi: 10.1016/j.jare.2021.08.003.
- Y. Hou et al., “NAD+ supplementation normalizes key Alzheimer’s features and DNA damage responses in a new AD mouse model with introduced DNA repair deficiency,” Proc Natl Acad Sci U S A, vol. 115, no. 8, pp. E1876–E1885, Feb. 2018, doi: 10.1073/pnas.1718819115.
- Q. Shen et al., “Biological synthesis of nicotinamide mononucleotide,” Biotechnol Lett, vol. 43, no. 12, pp. 2199–2208, Dec. 2021, doi: 10.1007/s10529-021-03191-1.
- W. Grabowska, E. Sikora, and A. Bielak-Zmijewska, “Sirtuins, a promising target in slowing down the ageing process,” Biogerontology, vol. 18, no. 4, pp. 447–476, 2017, doi: 10.1007/s10522-017-9685-9.
- G. F. S. Alegre and G. M. Pastore, “NAD+ Precursors Nicotinamide Mononucleotide (NMN) and Nicotinamide Riboside (NR): Potential Dietary Contribution to Health,” Curr Nutr Rep, vol. 12, no. 3, pp. 445–464, Sep. 2023, doi: 10.1007/s13668-023-00475-y.
- K. F. Mills et al., “Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice,” Cell Metab., vol. 24, no. 6, Art. no. 6, 13 2016, doi: 10.1016/j.cmet.2016.09.013.
- Y. Miao, Z. Cui, Q. Gao, R. Rui, and B. Xiong, “Nicotinamide Mononucleotide Supplementation Reverses the Declining Quality of Maternally Aged Oocytes,” Cell Rep, vol. 32, no. 5, p. 107987, Aug. 2020, doi: 10.1016/j.celrep.2020.107987.
- M. Kim, J. Seol, T. Sato, Y. Fukamizu, T. Sakurai, and T. Okura, “Effect of 12-Week Intake of Nicotinamide Mononucleotide on Sleep Quality, Fatigue, and Physical Performance in Older Japanese Adults: A Randomized, Double-Blind Placebo-Controlled Study,” Nutrients, vol. 14, no. 4, p. 755, Feb. 2022, doi: 10.3390/nu14040755.
- A. N. Long, K. Owens, A. E. Schlappal, T. Kristian, P. S. Fishman, and R. A. Schuh, “Effect of nicotinamide mononucleotide on brain mitochondrial respiratory deficits in an Alzheimer’s disease-relevant murine model,” BMC Neurol, vol. 15, p. 19, Mar. 2015, doi: 10.1186/s12883-015-0272-x.
- X. Wang, X. Hu, Y. Yang, T. Takata, and T. Sakurai, “Nicotinamide mononucleotide protects against β-amyloid oligomer-induced cognitive impairment and neuronal death,” Brain Res, vol. 1643, pp. 1–9, Jul. 2016, doi: 10.1016/j.brainres.2016.04.060.
- S. Tarantini et al., “Nicotinamide mononucleotide (NMN) supplementation rescues cerebromicrovascular endothelial function and neurovascular coupling responses and improves cognitive function in aged mice,” Redox Biol, vol. 24, Apr. 2019, doi: 10.1016/j.redox.2019.101192.
- J. Yoshino, K. F. Mills, M. J. Yoon, and S. Imai, “Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice,” Cell Metab, vol. 14, no. 4, pp. 528–536, Oct. 2011, doi: 10.1016/j.cmet.2011.08.014.
- M. Yoshino et al., “Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women,” Science, vol. 372, no. 6547, pp. 1224–1229, Jun. 2021, doi: 10.1126/science.abe9985.
- B. Zhu et al., “Levels of Fibrin Degradation Products at Admission With Acute Ischemic Stroke Correlate With the NIH Stroke Scale Score 1 h After Intravenous Thrombolysis,” Front. Neurol., vol. 12, May 2021, doi: 10.3389/fneur.2021.651867.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat, or cure any medical condition, ailment, or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.