Pancragen 20mg (Bioregulator)
$65.00
Pancragen is a peptide bioregulator derived from pancreatic tissue extracts studied for its role in modulating endocrine and exocrine gene expression. It influences peptide-mediated regulation of insulin synthesis and enzymatic activity through transcriptional and receptor-level signaling in preclinical models. Pancragen is used in research investigating pancreatic peptide signaling, metabolic regulation, and cellular homeostasis mechanisms.
For research use only. Not for human consumption.
References:
Khavinson VK et al., Bull Exp Biol Med, 2003 136(1):55–58
Anisimov VN et al., Biochemistry (Moscow), 2000 65(8):901–908
Morozov VG et al., Mech Ageing Dev, 1997 96(1–3):123–132
Overview
Pancragen is an endogenous short-chain tetrapeptide that has been described in the scientific literature as a pancreas-associated peptide bioregulator. In preclinical research contexts, peptides of this class are investigated for their capacity to modulate transcriptional and phenotypic markers associated with pancreatic cell state and differentiation. Reported experimental models include in-vitro cellular systems and in-vivo animal studies in which molecular and biochemical endpoints related to pancreatic endocrine and exocrine function are evaluated. Mechanistic interest has focused on peptide-mediated regulation of gene expression programs and chromatin-associated control of cellular function across defined experimental time courses.
Biochemical Characteristics
Amino Acid Sequence: Lys-Glu-Asp-Trp (KEDW)
Molecular Formula: C26H36N6O9
Molecular Weight: 576.25 g/mol
PubChem CID: 68452887
Synonyms: SCHEMBL5491754
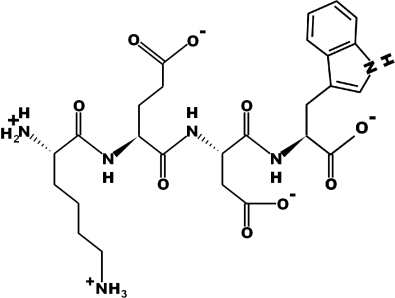 Source: PepDraw
Source: PepDraw
Research Applications
Pancragen is utilized in laboratory research to investigate peptide-mediated regulation of pancreatic cellular state and signaling behavior. Typical research-only applications include: (i) in-vitro studies examining gene expression, protein abundance, and proliferation or differentiation markers in pancreatic cell cultures; (ii) in-vivo animal studies assessing biochemical endpoints related to pancreatic endocrine output and systemic metabolic signaling; and (iii) exploratory epigenetic investigations evaluating peptide-associated modulation of transcriptional regulation across defined experimental durations.
Pathway / Mechanistic Context
Preclinical studies describe Pancragen as a regulatory signal capable of influencing transcriptional markers associated with pancreatic cell differentiation and functional state. Experimental analyses focus on changes in differentiation-associated factors within pancreatic-derived cellular systems, including markers characteristic of acinar and islet cell lineages. Endocrine-associated lineages are commonly evaluated using molecular and biochemical indicators related to insulin and glucagon signaling pathways within controlled experimental designs.
Diurnal and Endocrine Signaling Context
Several cited preclinical sources examine interactions between pancreatic endocrine signaling and diurnal regulatory systems via melatonin receptor expression in pancreatic islets. Within this mechanistic framework, receptor-mediated modulation of insulin and glucagon signaling is evaluated alongside circadian organization of metabolic pathways. Observed peptide-associated changes in endocrine biomarkers are discussed in relation to feedback interactions among pancreatic signaling networks and melatonin-associated pathways, without implication of translational or clinical relevance.
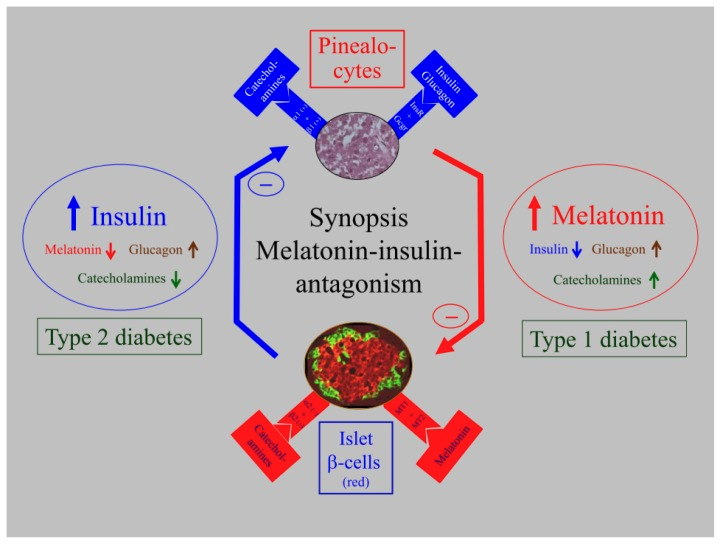 Source: PubChem
Source: PubChem
This schematic is reproduced from the scientific literature and is provided solely for mechanistic context. It does not imply human use, diagnosis, or therapeutic application.
Preclinical Research Summary
Across the cited literature, Pancragen is reported in cellular and animal models to correlate with changes in biochemical indices associated with pancreatic endocrine activity. Experimental studies in rodents and non-human primates describe modulation of glucose–insulin axis biomarkers under controlled laboratory conditions, with some investigations employing reference compounds solely as experimental comparators. Additional in-vitro research reports altered expression of molecular markers associated with extracellular matrix remodeling, cellular signaling, and cell state dynamics, including matrix metalloproteinases (MMP2, MMP9), selected signaling proteins, and proliferation-associated markers such as PCNA and Ki67. Concurrent observations include shifts in apoptosis-associated signaling proteins, interpreted within the literature as reflective of transcriptional and epigenetic regulatory mechanisms operating during defined experimental time courses.
Microvascular and Endothelial Endpoints in Animal Models
Certain rodent studies referenced evaluate microvascular parameters, including capillary permeability and endothelial adhesion, as downstream experimental readouts within metabolic dysregulation models. In these contexts, vascular endpoints are employed to characterize systemic effects of altered metabolic signaling in vivo and to assess correlations between peptide exposure and measured endothelial parameters under controlled laboratory conditions.
Form & Analytical Testing
This product is supplied strictly as a research reagent. Analytical characterization is performed to support identity and purity verification for laboratory use. Standard analytical methodologies for short peptides may include chromatographic purity assessment (e.g., HPLC or UPLC), molecular mass confirmation via mass spectrometry, and additional physicochemical analyses as appropriate. Researchers should reference the lot-specific certificate of analysis (COA) for detailed analytical data relevant to experimental documentation.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
 Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Prof. Vladimir Khavinson is being referenced as one of the leading scientists involved in the research and development of Pancragen. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide.
Referenced Citations
- V. K. Khavinson et al., “Effects of pancragen on the differentiation of pancreatic cells during their ageing,” Bull. Exp. Biol. Med., vol. 154, no. 4, pp. 501–504, Feb. 2013, doi: 10.1007/s10517-013-1987-6.
- O. V. Korkushko, V. K. Khavinson, V. B. Shatilo, I. A. Antonyk-Sheglova, and E. V. Bondarenko, “Prospects of using pancragen for correction of metabolic disorders in elderly people,” Bull. Exp. Biol. Med., vol. 151, no. 4, pp. 454–456, Aug. 2011, doi: 10.1007/s10517-011-1354-4.
- S. I. Rapoport, A. I. Molchanov, V. A. Golichenkov, O. V. Burlakova, E. S. Suprunenko, and E. S. Savchenko, “[Metabolic syndrome and melatonin],” Klin. Med. (Mosk.), vol. 91, no. 11, pp. 8–14, 2013.
- E. Peschke, I. Bähr, and E. Mühlbauer, “Melatonin and pancreatic islets: interrelationships between melatonin, insulin and glucagon,” Int. J. Mol. Sci., vol. 14, no. 4, pp. 6981–7015, Mar. 2013, doi: 10.3390/ijms14046981.
- N. D. Goncharova, L. G. Ivanova, T. E. Oganyan, A. A. Vengerin, and V. K. Khavinson, “[Correction of impaired glucose tolerance using tetrapeptide (Pancragen) in old female rhesus monkeys],” Adv. Gerontol. Uspekhi Gerontol., vol. 28, no. 3, pp. 579–585, 2015.
- V. K. Khavinson, M. M.-G. Gapparov, N. E. Sharanova, A. V. Vasilyev, and G. A. Ryzhak, “Study of biological activity of Lys-Glu-Asp-Trp-NH2 endogenous tetrapeptide,” Bull. Exp. Biol. Med., vol. 149, no. 3, pp. 351–353, Sep. 2010, doi: 10.1007/s10517-010-0944-x.
- N. D. Goncharova, L. G. Ivanova, T. É. Oganian, A. A. Vengerin, and V. K. Khavinson, “[Impact of tetrapeptide pancragen on endocrine function of the pancreas in old monkeys],” Adv. Gerontol. Uspekhi Gerontol., vol. 27, no. 4, pp. 662–667, 2014.
- I. M. Kvetnoi, A. P. Ryzhak, I. N. Kostyuchek, and Y. A. Tafeev, “Effect of tetrapeptide pancragene on functional morphology of the pancreas in rats with experimental diabetes mellitus,” Bull. Exp. Biol. Med., vol. 143, no. 3, pp. 368–371, Mar. 2007, doi: 10.1007/s10517-007-0114-y.
- V. K. Khavinson et al., “[Tetrapeptide stimulates functional activity of the pancreatic cells in aging],” Adv. Gerontol. Uspekhi Gerontol., vol. 25, no. 4, pp. 680–684, 2012.
- V. V. Ashapkin, N. S. Linkova, V. K. Khavinson, and B. F. Vanyushin, “Epigenetic mechanisms of peptidergic regulation of gene expression during aging of human cells,” Biochem. Biokhimiia, vol. 80, no. 3, pp. 310–322, Mar. 2015, doi: 10.1134/S0006297915030062.
- V. K. Khavinson, N. A. Gavrisheva, V. V. Malinin, S. G. Chefu, and E. L. Trofimov, “Effect of pancragen on blood glucose level, capillary permeability and adhesion in rats with experimental diabetes mellitus,” Bull. Exp. Biol. Med., vol. 144, no. 4, pp. 559–562, Oct. 2007, doi: 10.1007/s10517-007-0377-3.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.