PEG MGF 5mg
$95.00
PEG-MGF (PEGylated Mechano Growth Factor) is a PEGylated splice variant of IGF-1 (IGF-1Ec) designed to extend peptide stability and half-life for research applications. It acts on the IGF-1R and PI3K/Akt pathways, influencing muscle repair and cell proliferation signaling in vitro. PEG-MGF is used to model the effects of mechano-sensitive peptides on tissue regeneration and anabolic signaling cascades.
For research use only. Not for human consumption.
References: Yang SY et al., FEBS Lett, 1996 390(3):289–292
Philippou A et al., Mol Biol Rep, 2007 34(1):37–44
Mills P et al., Growth Horm IGF Res, 2007 17(5):376–385
Overview
PEG-MGF is a pegylated peptide format associated with IGF-1 Ec / mechano-growth factor (MGF) E-domain sequence biology. In laboratory research, PEG-MGF is used as a pathway probe to investigate IGF-axis signaling, myogenic program regulation in cellular models, and injury-response signaling networks in controlled in-vitro and in-vivo animal study designs. Pegylation is a chemical modification strategy that can be used to alter physicochemical behavior and extend peptide persistence in certain assay matrices and pharmacokinetic workflows relative to non-pegylated fragments.
Mechanistic studies of MGF-related sequences commonly evaluate receptor-linked signaling under defined exposure conditions, including comparative signaling readouts anchored to IGF-1 receptor (IGF-1R) activation and downstream pathway markers (e.g., PI3K/AKT and MAPK/ERK pathway panels) in preclinical systems.[4]
Biochemical Characteristics
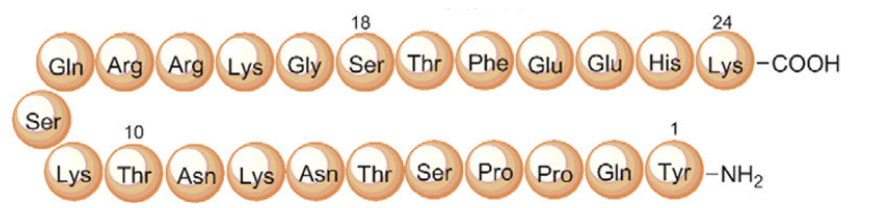 Primary sequence of MGF without polyethylene glycol attached
Primary sequence of MGF without polyethylene glycol attachedSource: ResearchGate
Sequence: PEG-Suc-Tyr-Gln-Pro-Pro-Ser-Thr-Asn-Lys-Asn-Thr-Lys-Ser-Gln-Arg-Arg-Lys-Gly-Ser-Thr-Phe-Glu-Glu-Arg-Lys-Cys
Molecular Formula: C121H200N42O39
PubChem SID: 178101669
Synonyms: Pegylated MGF, PEG IGF-1 Ec, PEG myotrophin
PEG-MGF is commonly described as an MGF-related peptide sequence presented in a pegylated format (often via PEG and linker chemistry such as succinylation) to modify solution behavior and persistence in experimental workflows. In peptide research, PEG conjugation is used to increase apparent hydrodynamic radius, alter adsorption behavior, and reduce clearance rates in certain in-vivo model designs, enabling time-course experiments that require longer effective exposure windows.
Research Applications
1. Skeletal Muscle Injury Models & Immune-Cell Dynamics
Preclinical studies have investigated MGF-related sequences in rodent skeletal muscle injury paradigms with endpoints that include inflammatory mediator expression, oxidative stress markers, and immune-cell recruitment/clearance kinetics within injured tissue.[1], [2] These experimental designs are used to map how IGF-1 isoform–linked signaling intersects with macrophage and neutrophil resolution programs and regeneration-associated gene expression profiles.
Related mechanistic literature characterizes exercise-associated expression patterns of IGF-1 isoforms and describes in-vitro assays evaluating MGF E-domain peptide inputs, supporting their use as pathway probes in muscle cell and tissue models.[3]
2. IGF-1 Receptor Signaling as a Mechanistic Anchor
In receptor-focused studies, full-length MGF has been evaluated for IGF-1R activation under controlled, equimolar exposure conditions, providing a comparative framework against recombinant IGF-1 in signaling assays.[4] These data support the use of MGF-related sequences as experimental inputs for mapping IGF-1R-driven pathway readouts (e.g., PI3K/AKT and MAPK/ERK panels) and downstream transcriptional signatures in preclinical systems.
3. Pegylated Formats for Time-Course & Distribution Studies
In time-dependent preclinical studies, short exposure windows can limit interpretation of non-pegylated peptide inputs. Pegylated formats (e.g., PEG-MGF) are therefore used in research designs that require longer circulating exposure relative to non-pegylated fragments, including single-dose time-course studies that track pathway activation and tissue distribution endpoints. Experimental discussion in the literature also notes delivery-route and localization constraints that motivate longer-persistence peptide formats for selected model designs.[5]
4. Cardiac Hypoxia/Ischemia Models & Cell-Death Signaling Endpoints
Bioengineering and biomaterials studies have explored localized MGF peptide delivery platforms in rodent cardiac hypoxia/ischemia models, measuring apoptosis-associated endpoints and recruitment of progenitor/stem-like cell populations to injury sites.[6] Additional work has examined polymeric microstructure delivery of MGF E-domain peptides in myocardial infarction models with hemodynamic and remodeling-associated readouts in controlled preclinical settings.[7]
5. Osteoblast Proliferation & Bone-Defect Models
Preclinical orthopedic research has evaluated MGF E-domain peptides in rabbit bone-defect models, focusing on osteoblast proliferation markers and comparative timelines under controlled study conditions.[8] Such work is used to investigate growth-factor axis modulation in mineralization and bone remodeling biology in animal systems.
6. Chondrocyte Migration & Mechanotransduction Pathways
Cell and tissue studies in mice have examined MGF-related peptide inputs in mechanical overload contexts, with mechanistic emphasis on RhoA/YAP-associated signaling and migration behavior of growth plate chondrocytes.[9] These models support use of MGF-derived peptides as probes for mechanotransduction-linked pathways in cartilage-relevant research.
7. Periodontal Ligament Cell Models & MEK/ERK Signaling
In-vitro studies using periodontal ligament cell cultures have investigated stretch-associated osteogenic differentiation programs and matrix metalloproteinase expression (e.g., MMP-1, MMP-2), with mechanistic focus on MEK/ERK1/2 pathway activation.[10]
8. CNS-Context Preclinical Literature
Preclinical reports and reviews have discussed MGF-related sequences in CNS contexts, including animal models of neuron injury and ischemia, with endpoints that include neuronal survival-associated markers and functional readouts measured in vivo.[12], [11]
Pathway / Mechanistic Context
MGF-related sequences are frequently studied within the broader IGF-axis network, using receptor-anchored assays to characterize signaling kinetics and downstream pathway modulation. In mechanistic workflows, IGF-1R engagement provides a reference framework for mapping intracellular signaling panels, including phosphorylation-state readouts and transcriptional programs associated with proliferation/differentiation processes in cell-based systems.[4]
Pegylation modifies peptide physicochemical properties and can extend the effective exposure window in selected experimental designs. For pathway mapping studies that rely on time-dependent readouts (e.g., early vs. late phosphorylation events, gene-expression trajectories, tissue distribution endpoints), pegylated formats can be used to reduce rapid loss of signal due to clearance or instability in certain matrices.
Preclinical Research Summary
Preclinical research involving MGF-related sequences includes in-vivo rodent injury paradigms (skeletal muscle and cardiac contexts), as well as in-vitro cell models (myoblasts, chondrocytes, periodontal ligament cells) used to measure pathway activity, gene-expression changes, and migration/proliferation-associated endpoints. Reported animal studies have evaluated inflammatory mediator profiles, immune-cell resolution kinetics, apoptosis-associated markers, and mechanotransduction-linked signaling readouts under controlled conditions.[1], [2], [6], [7], [9]
PEG-MGF is used in experimental designs where peptide persistence and time-course control are key variables, including studies that compare signaling trajectories across exposure durations and delivery formats.
Form & Analytical Testing
Product characterization in RUO workflows commonly includes identity confirmation and purity profiling using analytical techniques such as HPLC and mass spectrometry. When lot-specific documentation is provided, it is used for laboratory quality review (e.g., identity, purity, and consistency) within the experimental context.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Paul Goldspink, PhD is the Principal Investigator and Associate Professor at the Department of Physiology within the Medical College of Wisconsin. The research in Dr. Goldspink’s laboratory is focused on understanding the actions of IGF-1 isoforms in the heart and other tissues. We are investigating the role of IGF-1 isoforms in response to stresses such a mechanical overload, hypoxia, oxidative stress and age in the heart. We have focused on a particular isoform called Mechano-Growth Factor (MGF), which plays a protective role in preventing cell death, preserving contractility and preventing pathologic hypertrophy of the heart following myocardial infarction. We are currently investigating the underlying mechanisms utilizing peptide analogs derived from the E-domain region of MGF which serve as allosteric modulators of excitation-transcription pathways in muscle. Through collaborations we have been able to exploit our findings by developing a technology that combines a microscopic physical scaffold to deliver peptide therapeutics, with the goal of improving cardiac function during the progression of heart failure by using implantable cell-sized “biomimetic devices”. The development of these intelligent drug delivery platforms which we have recently employed in the heart, also have applicability of other tissues and disease states.
Paul Goldspink, PhD is being referenced as one of the leading scientists involved in the research and development of PEG-MGF. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Paul Goldspink, PhD is listed in [5] and [7] under the referenced citations.
Referenced Citations
- X. Liu, Z. Zeng, L. Zhao, P. Chen, and W. Xiao, “Impaired Skeletal Muscle Regeneration Induced by Macrophage Depletion Could Be Partly Ameliorated by MGF Injection,” Front. Physiol., vol. 10, p. 601, 2019.
- K.-T. Sun, K.-K. Cheung, S. W. N. Au, S. S. Yeung, and E. W. Yeung, “Overexpression of Mechano-Growth Factor Modulates Inflammatory Cytokine Expression and Macrophage Resolution in Skeletal Muscle Injury,” Front. Physiol., vol. 9, 2018.
- A. Philippou et al., “Expression of IGF-1 isoforms after exercise-induced muscle damage in humans: characterization of the MGF E peptide actions in vitro,” Vivo Athens Greece, vol. 23, no. 4, pp. 567–575, Aug. 2009.
- J. A. M. J. L. Janssen, L. J. Hofland, C. J. Strasburger, E. S. R. van den Dungen, and M. Thevis, “Potency of Full-Length MGF to Induce Maximal Activation of the IGF-I R Is Similar to Recombinant Human IGF-I at High Equimolar Concentrations,” PLoS ONE, vol. 11, no. 3, Mar. 2016.
- G. Goldspink, “Research on mechano growth factor: its potential for optimising physical training as well as misuse in doping,” Br. J. Sports Med., vol. 39, no. 11, pp. 787–788, Nov. 2005.
- G. Doroudian, J. Pinney, P. Ayala, T. Los, T. A. Desai, and B. Russell, “Sustained delivery of MGF peptide from microrods attracts stem cells and reduces apoptosis of myocytes,” Biomed. Microdevices, vol. 16, no. 5, pp. 705–715, Oct. 2014.
- J. R. Peña, J. R. Pinney, P. Ayala, T. A. Desai, and P. H. Goldspink, “Localized delivery of mechano-growth factor E-domain peptide via polymeric microstructures improves cardiac function following myocardial infarction,” Biomaterials, vol. 46, pp. 26–34, Apr. 2015.
- M. Deng et al., “Mechano growth factor E peptide promotes osteoblasts proliferation and bone-defect healing in rabbits,” Int. Orthop., vol. 35, no. 7, pp. 1099–1106, Jul. 2011.
- X. Jing et al., “Mechano-growth factor protects against mechanical overload induced damage and promotes migration of growth plate chondrocytes through RhoA/YAP pathway,” Exp. Cell Res., vol. 366, no. 2, pp. 81–91, May 2018.
- J.-T. Chen, Y. Wang, Z.-F. Zhou, and K.-W. Wei, “[Mechano-growth factor regulated cyclic stretch-induced osteogenic differentiation and MMP-1, MMP-2 expression in human periodontal ligament cells by activating the MEK/ERK1/2 pathway],” Shanghai Kou Qiang Yi Xue Shanghai J. Stomatol., vol. 28, no. 1, pp. 6–12, Feb. 2019.
- A. W., “Hearts and Minds of Mice and Men: Mechano Growth Factor a new tool in the battle against age-related neuron loss?,” On Biology, 20-Jul-2017.
- J. Dluzniewska et al., “A strong neuroprotective effect of the autonomous C-terminal peptide of IGF-1 Ec (MGF) in brain ischemia,” FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol., vol. 19, no. 13, pp. 1896–1898, Nov. 2005.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.