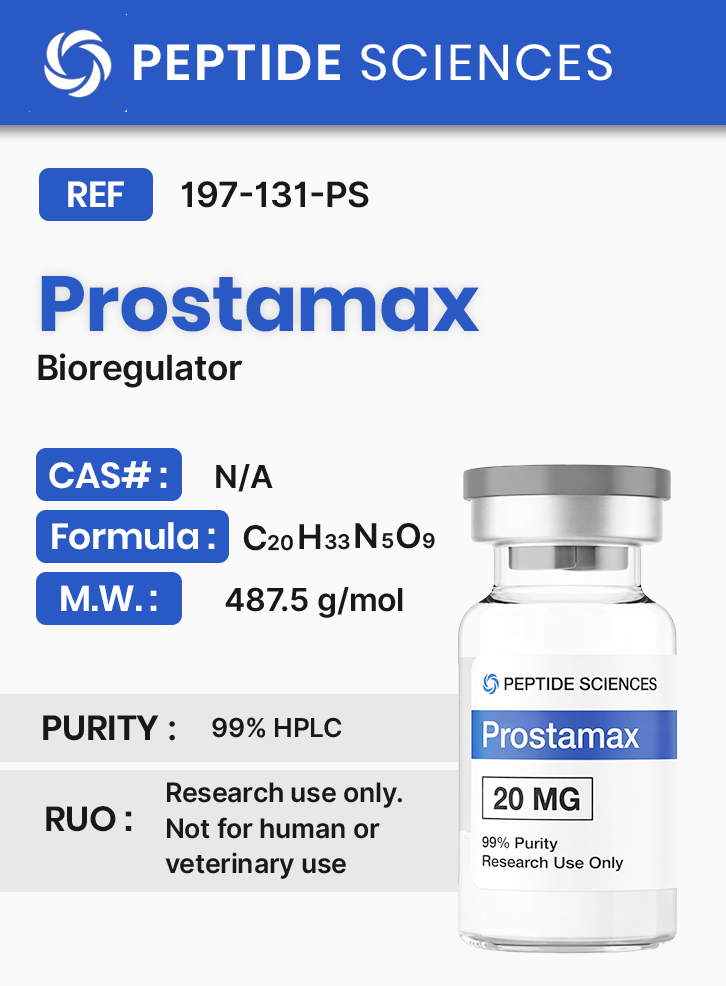
Prostamax 20mg (Bioregulator)
$75.00
Prostamax is a peptide bioregulator derived from prostate tissue extracts that modulates peptide-mediated regulation of cell differentiation and proliferation. Experimental studies indicate its effects on gene expression associated with epithelial integrity and endocrine feedback. Prostamax is used in molecular biology and reproductive research focusing on peptide regulation of glandular function.
For research use only. Not for human consumption.
References:
Linkova NS et al., Biogerontology, 2014;15(2):161–170
Morozov VG et al., Mech Ageing Dev, 1997;96(1–3):123–132
Anisimov VN et al., Arch Gerontol Geriatr, 2017;71:142–146
Overview
Prostamax is a synthetic short-chain tetrapeptide (Lys-Glu-Asp-Pro; KEDP) described in the scientific literature as a tissue-associated peptide bioregulator. In laboratory research settings, this peptide is investigated for its capacity to modulate chromatin organization, transcriptional accessibility, and ribosomal activity in organ-specific and immune-derived cellular systems. Experimental studies employ in-vitro cell cultures and in-vivo animal models to evaluate molecular, biochemical, and epigenetic endpoints under controlled conditions.
Biochemical Characteristics
Amino Acid Sequence: Lys-Glu-Asp-Pro (KEDP)
Molecular Formula: C20H33N5O9
Molecular Weight: 487.5 g/mol
PubChem CID: 9848296
Synonyms: SCHEMBL6660498
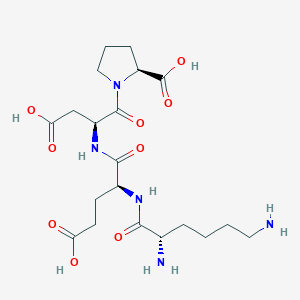 Source: PubChem
Source: PubChem
Research Applications
Prostamax is supplied strictly for laboratory research use. Documented experimental applications include organotypic tissue cultures, lymphocyte-based assays, and rodent models designed to examine peptide-mediated regulation of chromatin structure, ribosomal biogenesis, and transcriptional dynamics. Research use cases include analysis of heterochromatin accessibility, evaluation of immune-cell transcriptional readiness, and investigation of epigenetic regulation under experimentally induced stress or inflammatory conditions.
Pathway / Mechanistic Context
Mechanistic studies characterize Prostamax as an epigenetically active peptide capable of influencing higher-order chromatin organization. Experimental observations from cellular and animal models demonstrate peptide-associated decondensation of heterochromatin, resulting in increased accessibility of transcriptionally repressed genomic regions. These changes are evaluated alongside downstream alterations in transcriptional output and ribosome-associated protein synthesis.
Additional investigations focus on immune-derived cells, particularly lymphocytes, where Prostamax is reported to modulate ribosomal density and chromatin accessibility. These effects are assessed using molecular and cytogenetic techniques to characterize transcriptional and translational capacity under defined laboratory conditions.
Preclinical Research Summary
Preclinical investigations of Prostamax include in-vitro lymphocyte cultures, organ-specific tissue explants, and in-vivo rodent models. Within these experimental systems, the peptide is associated with observed changes in chromatin condensation, cellular composition, and tissue-level remodeling parameters. Animal studies evaluating experimentally induced inflammatory models report altered vascular and immune-cell markers following peptide exposure.
Comparative epigenetic studies further identify Prostamax-like peptides in long-lived rodent species, where they are examined in the context of lifespan-associated chromatin regulation. These findings are presented as mechanistic observations within experimental frameworks and do not imply translational or applied outcomes.
Form & Analytical Testing
This product is provided strictly as a laboratory research reagent. Analytical characterization may include chromatographic purity assessment (e.g., HPLC or UPLC) and molecular mass confirmation via mass spectrometry. Researchers should consult lot-specific certificates of analysis (COA) for detailed identity and purity information relevant to experimental documentation.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
 Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Vladimir Khavinson is a Professor, President of the European region of the International Association of Gerontology and Geriatrics; Member of the Russian and Ukrainian Academies of Medical Sciences; Main gerontologist of the Health Committee of the Government of Saint Petersburg, Russia; Director of the Saint Petersburg Institute of Bioregulation and Gerontology; Vice-president of Gerontological Society of the Russian Academy of Sciences; Head of the Chair of Gerontology and Geriatrics of the North-Western State Medical University, St-Petersburg; Colonel of medical service (USSR, Russia), retired. Vladimir Khavinson is known for the discovery, experimental and clinical studies of new classes of peptide bioregulators as well as for the development of bioregulating peptide therapy. He is engaged in studying of the role of peptides in regulation of the mechanisms of ageing. His main field of actions is design, pre-clinical and clinical studies of new peptide geroprotectors. A 40-year-long investigation resulted in a multitude of methods of application of peptide bioregulators to slow down the process of ageing and increase human life span. Six peptide-based pharmaceuticals and 64 peptide food supplements have been introduced into clinical practice by V. Khavinson. He is an author of 196 patents (Russian and international) as well as of 775 scientific publications. His major achievements are presented in two books: “Peptides and Ageing” (NEL, 2002) and “Gerontological aspects of genome peptide regulation” (Karger AG, 2005). Vladimir Khavinson introduced scientific specialty “Gerontology and Geriatrics” in the Russian Federation on the governmental level. Academic Council headed by V. Khavinson has oversighted over 200 Ph.D. and Doctorate theses from many different countries.
Prof. Vladimir Khavinson is being referenced as one of the leading scientists involved in the research and development of Prostamax. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide.
Referenced Citations
- A. N. Zakutskiĭ, N. I. Chalisova, G. A. Ryzhak, A. I. Aniskina, S. V. Filippov, and P. N. Zeziulin, “[The tissue-specific effect of synthetic peptides-biologic regulators in organotypic tissues culture in young and old rats],” Adv. Gerontol. Uspekhi Gerontol., vol. 19, pp. 93–96, 2006.
- T. G. Borovskaya et al., “Experimental studying of the drug efficiency Prostamax in the therapy of chronic aseptic prostatitis and its complications,” Mod. Res. Inflamm., vol. 2013, Jul. 2013, doi: 10.4236/mri.2013.23007.
- V. K. Khavinson, T. A. Lezhava, and V. V. Malinin, “Effects of short peptides on lymphocyte chromatin in senile subjects,” Bull. Exp. Biol. Med., vol. 137, no. 1, pp. 78–81, Jan. 2004, doi: 10.1023/b:bebm.0000024393.40560.05.
- T. A. Dzhokhadze, T. Z. Buadze, M. N. Gaĭozishvili, N. A. Baratashvili, and T. A. Lezhava, “[Deheterochromatinization of the chromatin in old age induced by oligopeptide bioregulator (]()_
- V. K. Khavinson, D. Y. Kormilets, and A. T. Mar’yanovich, “Peptides (Epigenetic Regulators) in the Structure of Rodents with a Long and Short Lifespan,” Bull. Exp. Biol. Med., vol. 163, no. 5, pp. 671–676, Sep. 2017, doi: 10.1007/s10517-017-3876-x.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.