Rigin 200mg (Palmitoyl Tetrapeptide-7) (Topical)
$200.00
Rigin (Palmitoyl Tetrapeptide-7) is a research peptide composed of Gly-Gln-Pro-Arg conjugated to palmitic acid, designed to modulate cytokine expression and dermal peptide signaling. It has been shown in preclinical research to influence extracellular matrix metabolism and cellular communication through interleukin and integrin pathways. Rigin is utilized in studies of peptide-mediated skin homeostasis and tissue remodeling.
For research use only. Not for human consumption.
References:
Katayama K et al., J Biochem, 2010 147(1):61–69
Pennefather PM et al., Exp Dermatol, 2012 21(7):512–518
Zhang L et al., Int J Cosmet Sci, 2020 42(3):302–310
Overview
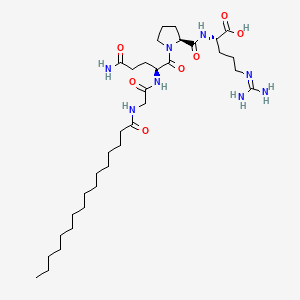
Rigin is used in the literature to describe either (i) an IgG-derived tetrapeptide fragment or (ii) the same tetrapeptide functionalized with a palmitoyl fatty-acid moiety (Pal-GQPR). In this description, “Rigin” refers to the palmitoyl-conjugated form (palmitoyl-Gly-Gln-Pro-Arg), a lipopeptide evaluated in preclinical systems as a tool to study cytokine-linked signaling and extracellular matrix (ECM) remodeling pathways under controlled laboratory conditions.
Biochemical Characteristics
Sequence (Three-Letter Code): palmitoyl-Gly-Gln-Pro-Arg-OH (Pal-GQPR)
Molecular formula: C34H62N8O7
Molecular Formula (Peptide Only): C18H32N8O6
Molecular weight: 694.9 g/mol
Synonyms: 221227-05-0, Palmitoyl tetrapeptide-7, N-Palmitoylrigin, Palmitoyl-GQPR, UNII-Q41S464P1R
The palmitoyl group increases overall hydrophobicity and is commonly used in peptide design to alter solubility and compartmental distribution in experimental systems. In RUO studies, palmitoylation is frequently evaluated for its impact on peptide–membrane interactions and cellular uptake in vitro.
Research Applications
- Use as a lipopeptide probe in preclinical models to evaluate cytokine-linked signaling networks (including IL-6–associated pathways) and downstream transcriptional responses.
- Investigation of ECM homeostasis and remodeling programs, including collagen/elastin-associated readouts and matrix turnover markers in cellular systems and in vivo animal studies.
- Evaluation of matrix metalloproteinase (MMP)–regulated ECM degradation processes using biochemical assays, gene expression endpoints, and protein-level markers in vitro.
- Comparative studies of tetrapeptide vs palmitoylated tetrapeptide to assess how lipidation alters physicochemical behavior and experimental performance in defined assay conditions.
Pathway / Mechanistic Context
Cytokine-linked signaling (IL-6 axis): Rigin (Pal-GQPR) has been described in preclinical literature as a modulator of inflammatory cytokine-associated signaling, including IL-6–linked responses. IL-6 functions as a pleiotropic signaling molecule that can regulate acute-phase and immune response gene programs in multiple cell types. In RUO studies, IL-6–associated endpoints are commonly used to quantify pathway activity and downstream effects on matrix-associated transcriptional outputs.
ECM remodeling and MMP-regulated matrix turnover: ECM integrity is governed by a balance between matrix synthesis (e.g., collagen/elastin deposition) and matrix degradation mediated in part by matrix metalloproteinases (MMPs). The cited literature discusses rigin in the context of MMP-associated activity and ECM turnover control in preclinical systems, which can be evaluated via MMP activity assays, ECM protein quantification, and expression profiling of remodeling markers.
IgG-fragment origin and peptide designation: The term “rigin” is used inconsistently across sources and may refer to the IgG-derived peptide fragment alone or the palmitoylated construct. For experimental clarity, RUO protocols should specify whether the test article is the tetrapeptide (GQPR) or the palmitoylated tetrapeptide (Pal-GQPR), as lipidation can materially change handling characteristics and assay behavior.
Preclinical Research Summary
Preclinical publications describe rigin (and/or Pal-GQPR) in experimental contexts that include immunomodulatory pathway assessment, cytokine-linked signaling readouts, and ECM remodeling endpoints in cell culture and animal models [2–4]. Additional reports discuss use in multi-ingredient research systems that pair palmitoylated peptides to explore combined effects on matrix-associated markers under controlled experimental conditions [3,5]. These findings should be interpreted strictly as preclinical observations for mechanistic investigation and assay design.
Form & Analytical Testing
Rigin (Palmitoyl Tetrapeptide-7; Pal-GQPR) is supplied as a research reagent intended for laboratory experimentation. Product identity and purity are typically confirmed using analytical methods such as HPLC and mass spectrometry (MS). Standard peptide handling practices should be used, and solvent/buffer selection should be optimized for lipopeptides due to increased hydrophobicity relative to non-lipidated peptides.
Referenced Citations
- Pubchem. Rigin | C18H32N8O6 – PubChem. Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Rigin#section=Top. (Accessed: 29th July 2016)
- Dutta, R. C., Puri, A. & Anand, N. Immunomodulatory potential of hydrophobic analogs of Rigin and their role in providing protection against Plasmodium berghei infection in mice. Int. Immunopharmacol. 1, 843–855 (2001).
- Mondon, P. et al. Evaluation of dermal extracellular matrix and epidermal-dermal junction modifications using matrix-assisted laser desorption/ionization mass spectrometric imaging, in vivo reflectance confocal microscopy, echography, and histology: effect of age and peptide applications. J. Cosmet. Dermatol. 14, 152–160 (2015).
- Sorsa, T., Tjäderhane, L. & Salo, T. Matrix metalloproteinases (MMPs) in oral diseases. Oral Dis. 10, 311–318 (2004).
- Matrixyl 3000 (palmitoyl oligopeptide & palmitoyl tetrapeptide-7): Back to the future of skin care. Available at: http://www.smartskincare.com/treatments/topical/palmitoyl-oligopeptide-palmitoyl-tetrapeptide-7-matrixyl-3000.html. (Accessed: 28th July 2016)
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.