Semax 30mg
$160.00
Semax is a synthetic heptapeptide (Met-Glu-His-Phe-Pro-Gly-Pro) derived from the ACTH(4-10) fragment. It is widely utilized in research investigating neurotrophic and nootropic signaling via modulation of BDNF, TrkB, and neurogenic transcription factors such as NGF and GAP-43. Experimental data indicate its capacity to affect redox balance, neuronal plasticity, and dopaminergic regulation in central nervous system models.
For research use only. Not for human consumption.
References:
Ashmarin IP et al., Neurosci Behav Physiol, 1998 28(3):341-348
Ashmarin IP et al., Neurochem J, 2005;39(1):36–44
Dolotov OV et al., Brain Res Bull, 2011 85(5):327-332
Overview
Semax (ACTH(4–7)-PGP; also referenced as Pro-Gly-Pro-ACTH in registry records) is a synthetic heptapeptide derived from a defined segment of adrenocorticotropic hormone (ACTH). In the scientific literature, Semax has been used as a research tool in neurobiology and molecular signaling studies, including investigations of activity-dependent gene programs, neurotrophin-linked transcriptional responses, monoaminergic pathway markers, and systems-level network activity measured with experimental imaging and omics approaches.
Preclinical publications describe Semax-associated changes in transcriptional signatures across CNS-relevant tissues and report modulation of neurotrophin-related gene expression (including BDNF and NGF) in controlled animal and cell-based experimental designs.
Biochemical Characteristics
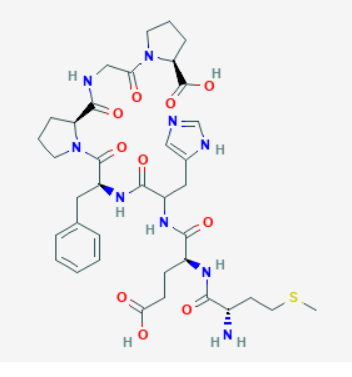 Image of Semax without the N-acetyl side chain
Image of Semax without the N-acetyl side chainSource: PubChem
Sequence: Met-Glu-His-Phe-Pro-Gly-Pro
Molecular Formula: C37H51N9O10S
Molecular Weight: 813.92 g/mol
CAS Number: 80714-61-0
Synonyms: Pro-Gly-Pro-ACTH
Research Applications
Semax is a synthetic analogue of an ACTH fragment, corresponding to amino acids 4–10 of ACTH with a C-terminal Pro-Gly-Pro motif. In laboratory research, Semax is used in preclinical models to probe pathway-level regulation of:
- Neurotrophin-linked transcription: experimental readouts involving BDNF and NGF gene-expression dynamics in CNS-relevant regions [5].
- Systems-level brain network activity: research imaging endpoints assessing resting-state network behavior and connectivity patterns [1].
- Neurovascular and immune-associated gene programs: genome-wide transcriptional profiling in animal models with emphasis on vascular-system and immune-system gene sets in brain tissue [3].
- Learning/memory model endpoints: mechanistic comparisons using ACTH-related peptides in rodent genetic models with cognition-focused behavioral readouts [6].
- Monoaminergic signaling markers: studies of serotonergic pathway measures and neurotrophin-dependent behavioral paradigms in rodents [7].
- Oxidative stress biomarker panels: liver-associated morphofunctional and lipid peroxidation endpoints under stress-model conditions [8] [9].
Preclinical Research Summary
Pathway / Mechanistic Context
Across cited preclinical literature, Semax is evaluated as a modulator of gene-expression programs and systems-level neural network activity. Reported endpoints include transcriptomic changes in CNS tissue, neurotrophin-associated transcriptional dynamics, and network-level activity patterns measured by experimental neuroimaging approaches. In addition, publications describe Semax-associated shifts in peripheral oxidative stress biomarker panels and hepatic tissue readouts under stress-model conditions.
1. Resting-State Network Readouts
Experimental imaging work reports changes in resting-state network behavior after Semax exposure, including measured effects on the default mode network as defined within that study’s analytic framework [1]. Background literature on default mode and social-cognition network relationships is commonly used to interpret these kinds of resting-state measures in neuroscience research [2].
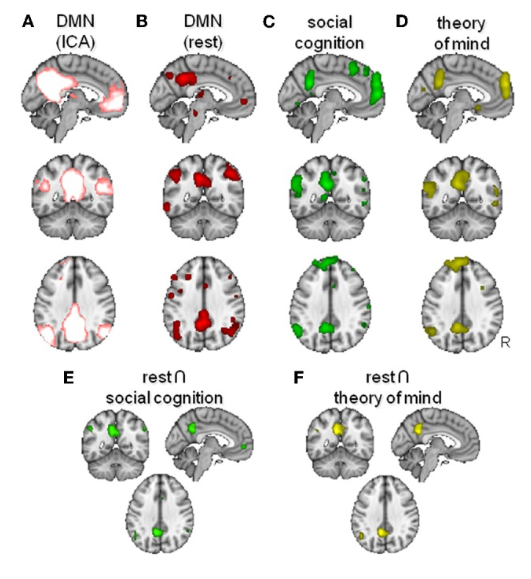 Image showing the overlap of activation between the resting mode network and parts of the brain responsible social cognition.
Image showing the overlap of activation between the resting mode network and parts of the brain responsible social cognition.
Source: PubMed
2. Genome-Wide Transcriptional Profiling in a Rodent Ischemia Model
In a rat model of focal brain ischemia, genome-wide transcriptional analysis reported Semax-associated changes in expression across immune- and vascular-system related gene sets in brain tissue, providing a molecular context for mechanistic investigation of neurovascular and inflammatory pathway regulation in this model [3].
3. Neurotrophin-Related Gene Expression Dynamics
Rodent studies report that Semax exposure is associated with time-dependent gene expression changes in the hippocampus and frontal cortex, including reported effects on BDNF and NGF gene-expression measures within the study design [5].
4. ACTH-Related Peptides in a Genetic Epilepsy Model
Preclinical work in a Kcna1-null mouse model reports that ACTH exposure is associated with preservation of learning and memory readouts in the experimental paradigm, providing comparative context for research on ACTH-derived peptide fragments such as Semax [6].
5. Serotonergic and Neurotrophin-Linked Behavioral Paradigms
Rodent studies evaluating altered hippocampal BDNF levels report behavioral and serotonergic consequences in experimental models, supporting broader mechanistic frameworks that connect neurotrophin abundance, serotonergic pathway markers, and behavioral outputs in preclinical research [7].
6. Peripheral Oxidative Stress and Hepatic Biomarker Readouts
Additional cited studies describe Semax-associated effects on hepatic morphofunctional endpoints and lipid peroxidation markers in rat stress-model conditions, including biochemical measures relevant to oxidative stress research workflows [8] [9].
Form & Analytical Testing
Semax is a synthetic peptide supplied for controlled laboratory workflows. Laboratories may reference sequence, CAS, and registry identifiers for internal documentation and study design. Analytical characterization for peptide materials commonly includes chromatographic purity profiling and mass confirmation consistent with internal qualification standards.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Igor Ivanovich Bobyntsev researches at the Kursk State Medical University within the Department of Pathophysiology. His main goal is to find new, more effective methods for teaching morphological disciplines… such as histology, cytology, embryology, human anatomy, pathological anatomy, the study of the morphological manifestation of stress-limiting effects of neuropeptides and their synthetic analogues. His key interests pertain to Antioxidants, Free Radicals, Antioxidant Activity, Free Radical Scavengers, Reactive Oxygen Species, Lipid Peroxidation, SOD, Oxidative Stress Biomarkers, Inflammatory Biomarkers, and Oxidative Stress. He specifically studied the influence of Semax on the morphofunctional state of hepatocytes, and lipid peroxidation in the liver, when under chronic emotional and painful stress.
Dr. Igor Ivanovich Bobyntsev is being referenced as one of the leading scientists involved in the research and development of Semax. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Igor Ivanovich Bobyntsev is listed in [8] and [9] under the referenced citations.
Referenced Citations
- I. S. Lebedeva et al., “Effects of Semax on the Default Mode Network of the Brain,” Bull. Exp. Biol. Med., vol. 165, no. 5, pp. 653–656, Sep. 2018. [PubMed]
- R. B. Mars, F.-X. Neubert, M. P. Noonan, J. Sallet, I. Toni, and M. F. S. Rushworth, “On the relationship between the ‘default mode network’ and the ‘social brain,’” Front. Hum. Neurosci., vol. 6, 2012. [PMC]
- E. V. Medvedeva et al., “The peptide semax affects the expression of genes related to the immune and vascular systems in rat brain focal ischemia: genome-wide transcriptional analysis,” BMC Genomics, vol. 15, p. 228, Mar. 2014. [PubMed]
- E. I. Gusev, M. Y. Martynov, E. V. Kostenko, L. V. Petrova, and S. N. Bobyreva, “[The efficacy of semax in the tretament of patients at different stages of ischemic stroke],” Zh. Nevrol. Psikhiatr. Im. S. S. Korsakova, vol. 118, no. 3. Vyp. 2, pp. 61–68, 2018. [PubMed]
- T. I. Agapova et al., “[Effect of semax on the temporary dynamics of brain-derived neurotrophic factor and nerve growth factor gene expression in the rat hippocampus and frontal cortex],” Mol. Genet. Mikrobiol. Virusol., no. 3, pp. 28–32, 2008. [PubMed]
- M. H. Scantlebury, K.-C. Chun, S.-C. Ma, J. M. Rho, and D. Y. Kim, “Adrenocorticotropic Hormone Protects Learning and Memory Function in Epileptic Kcna1-null mice,” Neurosci. Lett., vol. 645, pp. 14–18, Apr. 2017. [PubMed]
- T. Deltheil et al., “Behavioral and serotonergic consequences of decreasing or increasing hippocampus brain-derived neurotrophic factor protein levels in mice,” Neuropharmacology, vol. 55, no. 6, pp. 1006–1014, Nov. 2008. [PubMed]
- Ivanov, Alexander & Bobyntsev, Igor & Shepeleva, Olga & Kryukov, Alexey & Andreeva, L & Myasoedov, N. (2017). Influence of ACTG4-7-PGP (Semax) on Morphofunctional State of Hepatocytes in Chronic Emotional and Painful Stress. Bulletin of experimental biology and medicine. 163. [Research Gate]
- Bobyntsev, Igor & Kryukov, Alexey & Shepeleva, Olga & Ivanov, Alexander. (2015). The effect of ACTH-4-7-PGP peptide on lipid peroxidation in liver and activity of serum transaminases in rats under acute and chronic immobilization stress conditions. 78. 18-21. [Research Gate]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.