Sermorelin (5 mg)
$95.00
Sermorelin and Ipamorelin is a research peptide blend that combines a GHRH analog with a ghrelin receptor agonist to examine growth hormone regulation. Sermorelin (GHRH 1–29) activates the GHRH receptor to increase cAMP/PKA signaling, while Ipamorelin stimulates GHS-R1a through Gq/11-coupled calcium flux. This blend is employed in preclinical models investigating endocrine peptide synergy, GH release kinetics, and hypothalamic–pituitary signaling mechanisms.
For research use only. Not for human consumption.
References:
Bowers CY et al., Endocrinology, 1990 126(2):1165–1171
Frohman LA et al., Front Neuroendocrinol, 2002 23(3):257–291
Kojima M et al., Nature, 1999 402(6762):656–660
Overview
Sermorelin is a synthetic analogue of growth hormone–releasing hormone (GHRH) used in laboratory research to investigate signaling within the hypothalamic–pituitary somatotropic axis. In preclinical systems, GHRH-family peptides are applied to interrogate receptor binding, second-messenger signaling, and downstream transcriptional responses associated with growth hormone (GH) secretion dynamics and GH-linked signaling networks.
Experimental interest in sermorelin spans endocrine signaling, neuroendocrine regulation, and tissue-level remodeling signatures characterized in controlled in-vitro experiments and in-vivo animal models. All descriptions on this page are presented strictly for research contexts and do not imply any diagnostic, medical, or clinical use.
Biochemical Characteristics
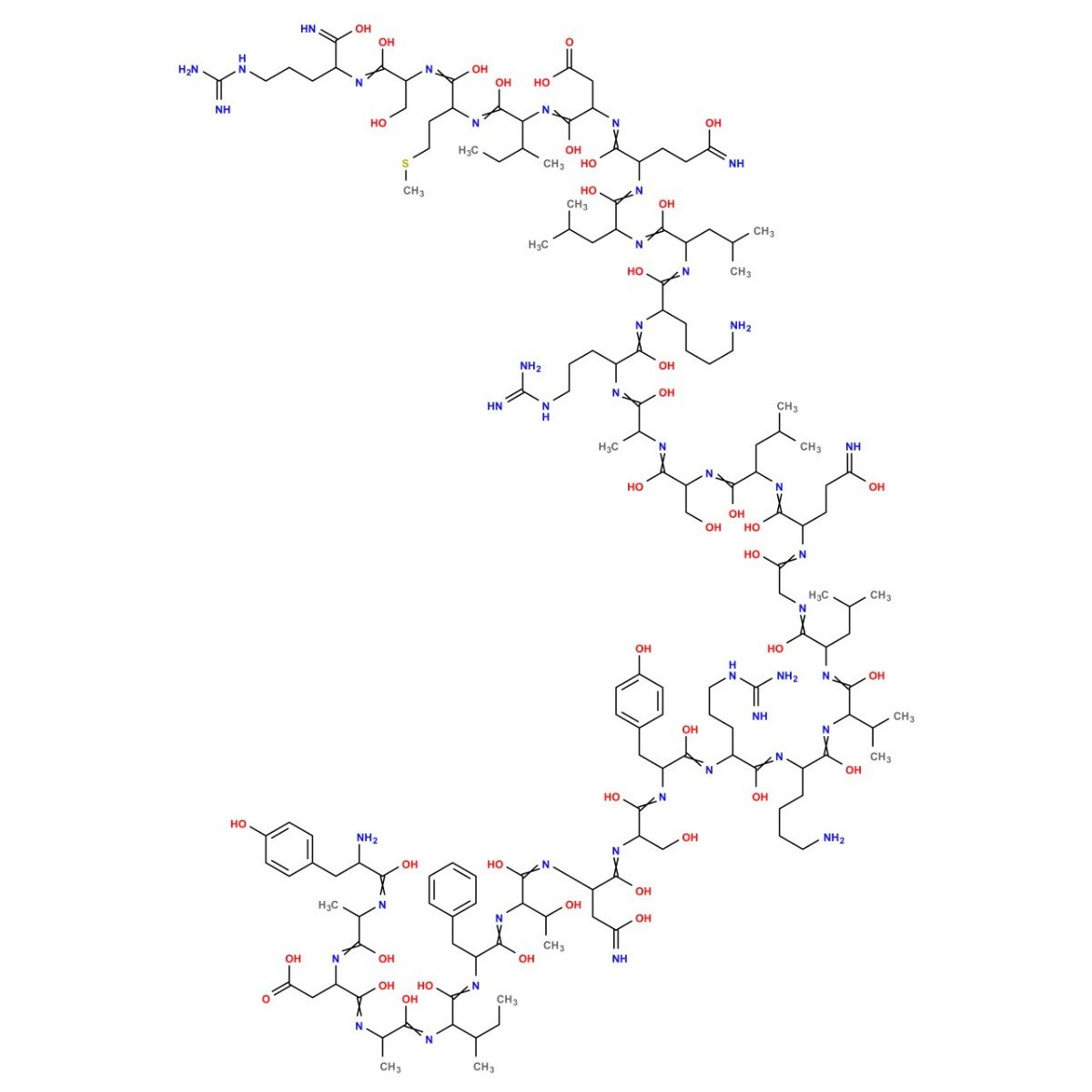 Sequence: Tyr-DL-Ala-DL-Asp-DL-Ala-DL-xiIle-DL-Phe-DL-xiThr-DL-Asn-DL-Ser-DL-Tyr-DL-Arg-DL-Lys-DL-Val-DL-Leu-Gly-DL-Gln-DL-Leu-DL-Ser-DL-Ala-DL-Arg-DL-Lys-DL-Leu-DL-Leu-DL-Gln-DL-Asp-DL-xiIle-DL-Met-DL-Ser-DL-Arg
Sequence: Tyr-DL-Ala-DL-Asp-DL-Ala-DL-xiIle-DL-Phe-DL-xiThr-DL-Asn-DL-Ser-DL-Tyr-DL-Arg-DL-Lys-DL-Val-DL-Leu-Gly-DL-Gln-DL-Leu-DL-Ser-DL-Ala-DL-Arg-DL-Lys-DL-Leu-DL-Leu-DL-Gln-DL-Asp-DL-xiIle-DL-Met-DL-Ser-DL-ArgMolecular Formula: C149H246N44O42S
Molecular Weight: 3357.933 g/mol
PubChem CID: 16129620
As a GHRH-family peptide analogue, sermorelin is used in biochemical workflows to evaluate ligand–receptor interactions, sequence–activity relationships, and stability/handling variables relevant to experimental design. Functional readouts are commonly generated in cell-based assays (e.g., receptor activation and downstream signaling) or in animal models where endocrine-axis activity can be quantified via established laboratory endpoints.
Research Applications
Sermorelin is supplied for research workflows that may include (non-exhaustive):
- GHRH receptor (GHRHR) pharmacology, including ligand binding and receptor-selectivity studies
- cAMP/PKA signaling and downstream transcriptional regulation in somatotroph-derived or engineered receptor-expression systems
- GH/IGF-axis pathway mapping in controlled preclinical models (molecular and tissue-level endpoints)
- Neuroendocrine signaling studies assessing hypothalamic peptide networks and sleep/feeding-related signaling mediators
- Exploratory in-vivo model work evaluating remodeling signatures (e.g., extracellular matrix turnover, angiogenesis markers, and inflammatory mediator profiles) under controlled experimental conditions
Pathway / Mechanistic Context
Mechanistically, GHRH-family ligands signal through the growth hormone–releasing hormone receptor (GHRHR), a class B G protein–coupled receptor. In canonical models, receptor engagement is associated with activation of adenylate cyclase and elevation of intracellular cAMP, supporting PKA-dependent phosphorylation events and regulated gene expression programs relevant to somatotroph function.
Downstream signaling contexts frequently evaluated in preclinical systems include pathway cross-talk with MAPK/ERK and PI3K/Akt signaling nodes, which are commonly studied for their roles in cell survival, differentiation programs, and stimulus-dependent transcriptional outputs. In tissue models, downstream readouts may include extracellular matrix (ECM) turnover markers, vascular remodeling/angiogenesis signatures, and cytokine/chemokine profiles measured using standard laboratory assays.
Preclinical Research Summary
GHRH-Analogue Signaling in Cardiac Remodeling Models
Preclinical large-animal studies have evaluated GHRH-analogue signaling in controlled ischemic injury models. Reported experimental endpoints include remodeling-associated measures such as apoptosis-associated markers, extracellular matrix organization, and microvascular/capillary density in peri-injury tissue regions, along with inflammatory mediator profiling in myocardial tissue[1], [2].
These investigations are typically used to inform mechanistic remodeling biology experiments (e.g., ECM deposition/turnover, angiogenesis-related gene expression, and inflammatory signaling changes) in vitro and in vivo, and are presented here solely as preclinical pathway context.
GHRH-Analogue Activity in Experimental Seizure Models
Rodent epilepsy models have been used to explore interactions between GHRH analogues and inhibitory neurotransmission systems. Published work has examined relationships between GHRH-family signaling and GABA receptor biology in brain tissue and experimental seizure paradigms, supporting mechanistic evaluation of receptor cross-talk in neurophysiological signaling networks[3].
Neuroendocrine Axis Coupling in Sleep-Related Signaling
Sleep–wake regulatory pathways are frequently investigated through neuropeptide systems, including orexin/hypocretin networks. In fish models, studies have assessed endocrine coupling between GHRH-axis activity and orexigenic signaling, providing a basis for mechanistic experiments examining neuroendocrine coordination and peptide–receptor signaling integration under controlled laboratory conditions[4].
Receptor Regulation and Desensitization Concepts in Peptide Signaling
Experimental frameworks for peptide ligands frequently assess receptor regulation phenomena (e.g., desensitization, internalization, and signaling bias) using established in-vitro models and systems-level readouts. Literature discussing receptor response dynamics and tachyphylaxis/desensitization concepts is frequently used to inform receptor pharmacology experiment design and interpretation of longitudinal signaling measurements in controlled settings[5], [6].
All research summaries above reflect preclinical and laboratory observations and are provided solely to support experimental planning and mechanistic discussion.
Form & Analytical Testing
Sermorelin supplied for research is typically prepared as a synthetic peptide for laboratory handling and downstream analytical verification. Product identity and purity are commonly confirmed using chromatographic and mass spectrometric methods. Batch-specific analytical documentation is provided below.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
 Richard F. Walker, Ph.D, R.Ph, lead author of A better approach to management of adult-onset growth hormone insufficiency?”, received a BS in pharmacy from Rutgers University, a MS in Biochemistry from New Mexico State University and a PhD in a physiology from Rutgers University. He holds postdoctoral fellowships in neuroendocrinology and neuropharmacology at Duke University College of Medicine (Center for the Study of Aging and Human Development) and the University of California, Berkeley, respectively.
Richard F. Walker, Ph.D, R.Ph, lead author of A better approach to management of adult-onset growth hormone insufficiency?”, received a BS in pharmacy from Rutgers University, a MS in Biochemistry from New Mexico State University and a PhD in a physiology from Rutgers University. He holds postdoctoral fellowships in neuroendocrinology and neuropharmacology at Duke University College of Medicine (Center for the Study of Aging and Human Development) and the University of California, Berkeley, respectively.
Richard F. Walker, Ph.D, R.Ph is being referenced for academic attribution purposes. No endorsement, affiliation, or commercial relationship is implied or expressed between any researcher and any product offering. The purpose of citing the author is to acknowledge published scientific contributions. Richard F. Walker, Ph.D, R.Ph is listed in [5] under the referenced citations.
Referenced Citations
- L. L. Bagno et al., “Growth Hormone–Releasing Hormone Agonists Reduce Myocardial Infarct Scar in Swine With Subacute Ischemic Cardiomyopathy,” J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis., vol. 4, no. 4, Mar. 2015.
- R. M. Kanashiro-Takeuchi et al., “New therapeutic approach to heart failure due to myocardial infarction based on targeting growth hormone-releasing hormone receptor,” Oncotarget, vol. 6, no. 12, pp. 9728–9739, Mar. 2015.
- S. Tang et al., “Interactions between GHRH and GABAARs in the brains of patients with epilepsy and in animal models of epilepsy,” Sci. Rep., vol. 7, Dec. 2017.
- B. S. Shepherd et al., “Endocrine and orexigenic actions of growth hormone secretagogues in rainbow trout (Oncorhynchus mykiss),” Comp. Biochem. Physiol. A. Mol. Integr. Physiol., vol. 146, no. 3, pp. 390–399, Mar. 2007.
- R. F. Walker, “Sermorelin: A better approach to management of adult-onset growth hormone insufficiency?,” Clin. Interv. Aging, vol. 1, no. 4, pp. 307–308, Dec. 2006.
- S. T. Wahid, P. Marbach, B. Stolz, M. Miller, R. A. James, and S. G. Ball, “Partial tachyphylaxis to somatostatin (SST) analogues in a patient with acromegaly: the role of SST receptor desensitisation and circulating antibodies to SST analogues,” Eur. J. Endocrinol., vol. 146, no. 3, pp. 295–302, Mar. 2002.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.







