TB-500 Fragment (17-23) 10mg
$100.00
TB-500 Fragment (17-23) is a research peptide consisting of a short amino acid sequence derived from the actin-binding region of Thymosin Beta-4. It has been shown in preclinical studies to regulate cell migration, angiogenesis, and cytoskeletal organization through modulation of actin polymerization. This fragment is used in experimental research investigating wound repair, tissue remodeling, and peptide-mediated regenerative signaling mechanisms.
For research use only. Not for human consumption.
References:
Goldstein AL et al., Ann NY Acad Sci, 2007 1112:148–154
Huff T et al., J Mol Biol, 2001 312(3):593–606
Malinda KM et al., FASEB J, 1999 13(14):2106–2114
TB-500 Fragment (17-23), also called fequesetide or (17) (LKKTETQ) (23), represents the smallest portion of the thymosin beta-4 molecule that retains the larger protein’s active binding domain. Research indicates that this synthetic derivative of thymosin beta-4 is capable of binding to actin, the molecule inside of cells that is responsible for improved cell structure, movement (a.k.a. migration), and replication. By altering the function of actin in cells, TB-500 Fragment (17-23) has been shown to modulate the immune response and alter cell migration patterns. This can lead to large-scale changes in tissue/organ structure and function. In animal models, these changes have been shown to accelerate wound healing, decrease inflammation, promote blood vessel growth, reduce scar formation, improve musculoskeletal function, and help to slow or reverse the course of certain disease conditions.
Overview
Thymosin Beta-4 Fragment (17–23), also known as fequesetide (LKKTETQ), is a synthetic heptapeptide corresponding to a conserved actin-interaction domain derived from the larger thymosin beta-4 polypeptide. This fragment is utilized in laboratory research settings as a molecular probe for investigating cytoskeletal organization, actin dynamics, and intracellular signaling processes.
All observations associated with this peptide originate from in-vitro systems and controlled in-vivo animal models. No clinical, diagnostic, or therapeutic interpretations are implied.
Biochemical Characteristics
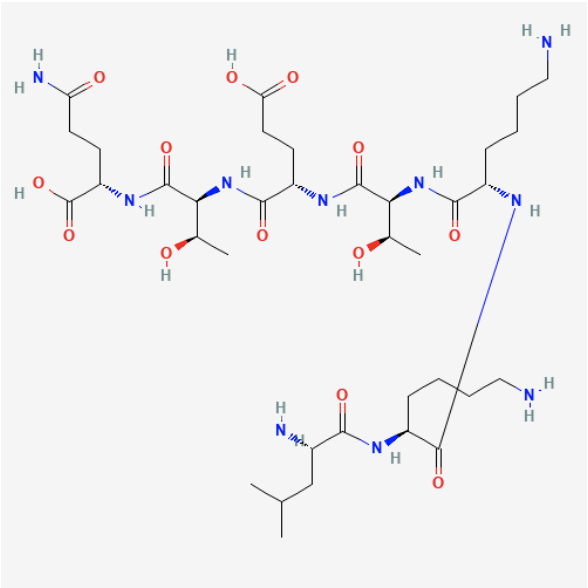
Amino Acid Sequence: LEU-LYS-LYS-THR-GLU-THR-GLN (LKKTETQ)
Chemical Formula: C36H66N10O13
Molecular Weight: 846.97 g/mol
PubChem CID: 10169788
CAS Number: 476014-70-7
Synonyms: Fequesetide; Thymosin Beta-4 (17–23)
The peptide lacks disulfide bonds and exhibits physicochemical properties suitable for aqueous laboratory systems. Structural simplicity enables reproducible synthesis and analytical verification.
Research Applications
Thymosin Beta-4 Fragment (17–23) is employed exclusively in research environments to examine:
- Actin monomer sequestration and filament assembly
- Cellular migration and cytoskeletal remodeling
- Intracellular signal transduction pathways
- Protein–protein interaction dynamics
- Regulation of inflammatory mediators in animal models
All applications are limited to non-clinical experimental models.
Pathway / Mechanistic Context
Mechanistic investigations indicate that the fragment participates in modulation of actin polymerization processes. Interaction with actin-regulatory complexes such as Arp2/3 has been explored in preclinical models, highlighting its utility for studying branched actin network formation.
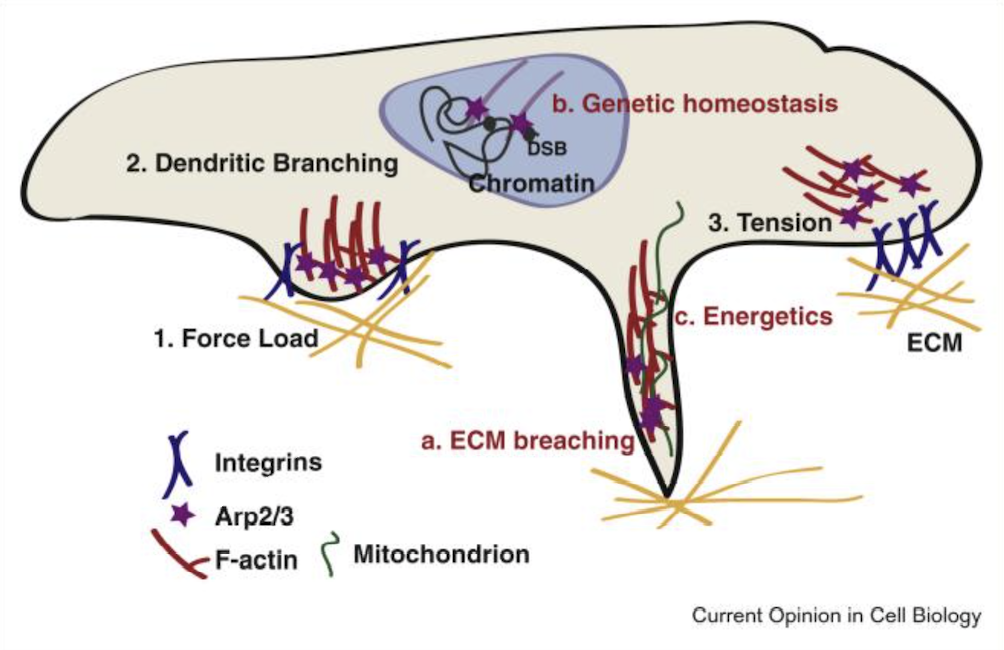
Downstream signaling pathways associated with cytoskeletal organization, including focal adhesion kinase–related cascades and PI3K/Akt-associated signaling, have been examined in vitro using this peptide as a research tool.
Preclinical Research Summary
Published preclinical literature describes investigation of thymosin beta-4–derived fragments in rodent and cellular systems. Experimental observations include modulation of fibroblast migration, endothelial cell organization, and cytokine signaling under controlled laboratory conditions.
Additional studies in neural and musculoskeletal animal models have explored molecular responses related to cytoskeletal plasticity, inflammatory mediator balance, and cellular differentiation markers. These findings are presented strictly as experimental observations without clinical extrapolation.
Form & Analytical Testing
This product is supplied as a lyophilized synthetic peptide produced via solid-phase peptide synthesis. Each batch is characterized using high-performance liquid chromatography and mass spectrometry to confirm identity and purity.
Analytical documentation supports reproducibility for laboratory workflows.
About The Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Geoffrey M. Cooper is professor of biology at Boston University. He served as chair of the department of biology for a number of years, and subsequently as associate dean of the faculty for the natural sciences in the university’s college of arts & sciences.
Cooper earned his Ph.D. at the University of Miami in 1973, and was a postdoctoral fellow with nobel laureate, Howard Temin. His work includes cellular growth control, cancer, and signal transduction. More specifically, he focuses on “the roles of proto-oncogene proteins as elements of signal transduction pathways that control proliferation, differentiation, and survival of mammalian cells.”
Geoffrey M. Cooper is being referenced as one of the leading scientists involved in the research and development of TB-500 Fragment (17-23). In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Geoffrey M. Cooper is listed in [2] under the referenced citations.
Resources
- V. Papalazarou and L. M. Machesky, “The cell pushes back: The Arp2/3 complex is a key orchestrator of cellular responses to environmental forces,” Curr. Opin. Cell Biol., vol. 68, pp. 37–44, Feb. 2021, doi: 10.1016/j.ceb.2020.08.012.
- G. M. Cooper, “The Cytoskeleton and Cell Movement,” in The Cell: A Molecular Approach. 2nd edition, Sinauer Associates, 2000. Accessed: Apr. 10, 2024. [Online]. Available: https://www.ncbi.nlm.nih.gov/books/NBK9893/
- “18.1: Introduction,” Biology LibreTexts. Accessed: Apr. 10, 2024. [Online]. Available: https://bio.libretexts.org/Under_Construction/Cell_and_Molecular_Biology_(Bergtrom)/18%3A_The_Cytoskeleton_and_Cell_Motility/18.01%3A_Introduction
- PubChem, “Lkktetq.” Accessed: Apr. 10, 2024. [Online]. Available: https://pubchem.ncbi.nlm.nih.gov/compound/10169788
- S. Lv, H. Cai, Y. Xu, J. Dai, X. Rong, and L. Zheng, “Thymosin-β 4 induces angiogenesis in critical limb ischemia mice via regulating Notch/NF-κB pathway,” Int. J. Mol. Med., vol. 46, no. 4, pp. 1347–1358, Oct. 2020, doi: 10.3892/ijmm.2020.4701.
- G. Sosne, P. Qiu, and M. Kurpakus-Wheater, “Thymosin beta-4 and the eye: I can see clearly now the pain is gone,” Ann. N. Y. Acad. Sci., vol. 1112, pp. 114–122, Sep. 2007, doi: 10.1196/annals.1415.004.
- S. S. Iyer and G. Cheng, “Role of Interleukin 10 Transcriptional Regulation in Inflammation and Autoimmune Disease,” Crit. Rev. Immunol., vol. 32, no. 1, pp. 23–63, 2012.
- A. Carracedo and P. P. Pandolfi, “The PTEN-PI3K pathway: of feedbacks and cross-talks,” Oncogene, vol. 27, no. 41, pp. 5527–5541, Sep. 2008, doi: 10.1038/onc.2008.247.
- G. Song, G. Ouyang, and S. Bao, “The activation of Akt/PKB signaling pathway and cell survival,” J. Cell. Mol. Med., vol. 9, no. 1, pp. 59–71, Jan. 2005, doi: 10.1111/j.1582-4934.2005.tb00337.x.
- J. P. Alao, “The regulation of cyclin D1 degradation: roles in cancer development and the potential for therapeutic invention,” Mol. Cancer, vol. 6, p. 24, Apr. 2007, doi: 10.1186/1476-4598-6-24.
- Y. Xiong et al., “Neuroprotective and neurorestorative effects of thymosin β4 treatment following experimental traumatic brain injury,” Ann. N. Y. Acad. Sci., vol. 1270, pp. 51–58, Oct. 2012, doi: 10.1111/j.1749-6632.2012.06683.x.
- D. C. Morris et al., “A dose-response study of thymosin β4 for the treatment of acute stroke,” J. Neurol. Sci., vol. 345, no. 1–2, pp. 61–67, Oct. 2014, doi: 10.1016/j.jns.2014.07.006.
- J. Zhang et al., “Thymosin beta4 promotes oligodendrogenesis in the demyelinating central nervous system,” Neurobiol. Dis., vol. 88, pp. 85–95, Apr. 2016, doi: 10.1016/j.nbd.2016.01.010.
- M. Santra et al., “Thymosin β4 up-regulation of microRNA-146a promotes oligodendrocyte differentiation and suppression of the Toll-like proinflammatory pathway,” J. Biol. Chem., vol. 289, no. 28, pp. 19508–19518, Jul. 2014, doi: 10.1074/jbc.M113.529966.
- M. Severa et al., “Thymosins in multiple sclerosis and its experimental models: moving from basic to clinical application,” Mult. Scler. Relat. Disord., vol. 27, pp. 52–60, Jan. 2019, doi: 10.1016/j.msard.2018.09.035.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.