Triptorelin (GnRH) 2mg
$42.00
Triptorelin is a synthetic decapeptide analog of gonadotropin-releasing hormone (GnRH; pGlu-His-Trp-Ser-Tyr-D-Trp-Leu-Arg-Pro-Gly-NH₂). It binds to the GnRH receptor, initiating Gq/PLC and IP₃–Ca²⁺ signaling cascades that regulate luteinizing and follicle-stimulating hormone release in experimental systems. Triptorelin is used in receptor biology and endocrine regulation research to study hypothalamic–pituitary–gonadal axis modulation.
For research use only. Not for human consumption.
References:
Sandow J et al., Life Sci, 1984 35(15):1503–1513
Conn PM et al., Endocr Rev, 1994 15(6): 682–715
Limonta P et al., J Steroid Biochem Mol Biol, 2003 85(2–5): 249–258
Overview
Triptorelin is a synthetic gonadotropin-releasing hormone (GnRH) analog (also referenced as an LHRH analog) used in research to study pituitary regulation of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), and downstream sex-hormone signaling. In clinical contexts, continuous long-term exposure is used to suppress testosterone and estrogen production as part of androgen deprivation therapy (notably in advanced prostate cancer) and for ovarian function suppression in certain hormone-sensitive settings.
Important research note: Triptorelin’s observed effects depend heavily on administration pattern (pulsatile vs. continuous exposure), which is why dosing protocol design is a central focus in endocrine and oncology research models.
Biochemical Characteristics
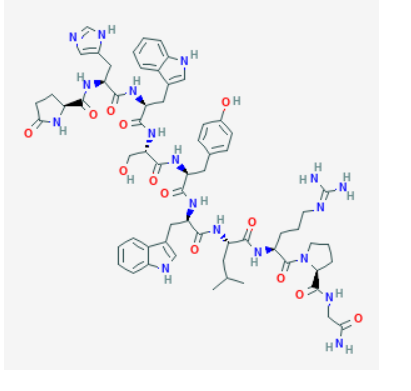 Source: PubChem
Source: PubChem
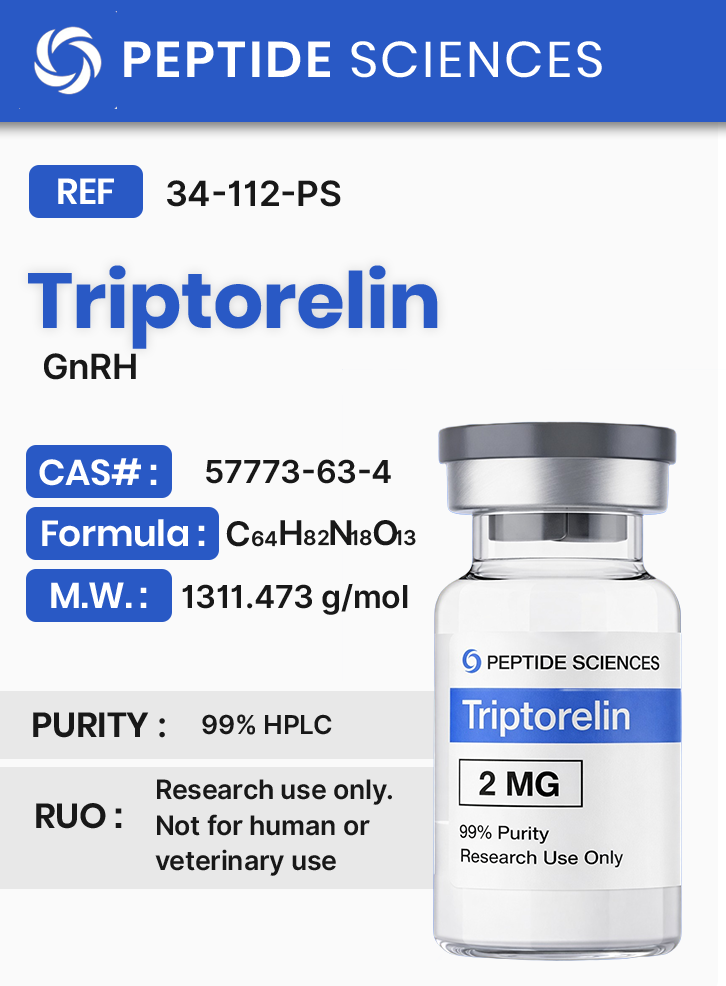
Sequence: Pyr-His-Trp-Ser-Tyr-D-Trp-Leu-Arg-Pro-Gly
Molecular Formula: C64H82N18O13
Molecular Weight: 1311.473 g/mol
PubChem CID: 25074470
CAS Number: 57773-63-4
Synonyms: Decapeptyl, TRP(6)-LHRH, Trelstar, Triptoreline, Gonapeptyl
Triptorelin is a decapeptide designed to mimic endogenous GnRH while incorporating an amino-acid substitution (D-Trp at position 6) that can alter receptor interaction dynamics and metabolic stability in experimental systems. In laboratory workflows, triptorelin is commonly characterized using chromatographic and spectrometric methods to confirm identity and purity.
Research Applications
Triptorelin is supplied for laboratory research and is commonly utilized as a mechanistic probe in controlled non-clinical studies involving:
- GnRH receptor (GnRHR) activation, desensitization, and internalization dynamics under pulsatile vs. continuous exposure paradigms
- LH/FSH secretion modeling and endocrine feedback-loop investigations in preclinical systems
- Hormone-trajectory studies relevant to androgen deprivation and ovarian suppression research frameworks
- Comparative endocrine pharmacology vs. other GnRH analogs (agonists/antagonists) in receptor pharmacology experiments
- Exploratory translational literature reviews in oncology and reproductive endocrinology (non-product-claims; background only)
All applications are restricted to controlled in-vitro and in-vivo animal research contexts and are not intended for diagnostic or therapeutic use.
Pathway / Mechanistic Context
Triptorelin primarily acts through the gonadotropin-releasing hormone receptor (GnRHR), a G protein–coupled receptor expressed in anterior pituitary gonadotrophs. In canonical models, receptor activation is linked to phospholipase C signaling, inositol phosphate generation, and intracellular calcium mobilization, which together support regulated secretion of LH and FSH.
A defining research feature of GnRH agonists is the strong dependence on temporal exposure pattern: pulsatile stimulation tends to support gonadotropin secretion, whereas continuous exposure can drive receptor desensitization/downregulation and suppression of LH/FSH, with downstream reduction in sex-hormone production. This pulse-versus-continuous distinction is a core design variable in endocrine, reproductive, and oncology research models.
Preclinical Research Summary
1. Protocol-Dependent Outcomes (Stimulation vs. Suppression)
Research and clinical literature consistently emphasizes that GnRH agonist outcomes are protocol-dependent: early-phase gonadotropin and testosterone increases can occur before suppression with sustained exposure (often described as a flare phenomenon). These dynamics inform endocrine modeling and timing-sensitive study designs in non-clinical and clinical-adjacent research contexts.
2. Oncology-Adjacent Research Contexts (Breast & Prostate)
Triptorelin is widely discussed in the literature in connection with hormone suppression strategies in oncology, including ovarian function suppression in certain breast cancer regimens and androgen deprivation therapy in prostate cancer settings [2], [3], [4]. These sources are provided as background references and do not imply product claims for any experimental use.
3. Reproductive Endocrinology and Gynecologic Research
Additional referenced literature discusses triptorelin in fertility-preservation contexts during chemotherapy and in gynecologic research areas such as endometriosis and adenomyosis, supporting broader investigation of GnRH-axis modulation in controlled research settings [8], [9], [12].
4. Endocrine–Immune Interaction (Preclinical)
Preclinical work has explored LHRH/GnRH agonist signaling in relation to thymic parameters and aging-associated immune changes, including thymus weight and thymocyte proliferative measures in experimental models [14].
Triptorelin exhibits moderate side effects, low oral and excellent subcutaneous bioavailability in mice. Per kg dosage in mice does not scale to humans. Triptorelin for sale is limited to educational and scientific research only, not for human consumption. Only buy Triptorelin if you are a licensed researcher.
Form & Analytical Testing
Triptorelin is typically supplied as a synthetic peptide intended for laboratory handling and controlled experimentation. Identity and purity are commonly verified using high-performance liquid chromatography (HPLC) and mass spectrometry (MS). Batch-specific analytical documentation supports experimental reproducibility and consistency.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Prof. Dr. Axel S. Merseburger is the chairman of the Department of Urology, Campus Lübeck, University Hospital Schleswig-Holstein, Germany. Professor Merseburger obtained his medical degree from Hannover Medical School in 2002 after a one-year academic research programme in Washington DC, USA, where he completed his MD thesis. In 2008, he accepted a staff position in Hannover and was promoted to associate professor in 2009; he obtained full professorship in 2012. Professor Merseburger is board certified in urology and is a member of numerous national and international urological and oncological associations. In addition to serving as a reviewer and member of the editorial board of many journals, Professor Merseburger is the associate editor of the World Journal of Urology and Editor-in-Chief of the “Advanced Prostate Cancer Resource Centre”. Part of his studies pertained to the examination of Triptorelin’s ability to induce androgen deprivation in order to treat prostate cancer. Furthermore, he is a member of the European Association of Urology (EAU) Guideline Groups on Renal Cell Cancer. He serves as national and international principal investigator in several phase II/III clinical trials. Professor Merseburger has won multiple awards, stipends and prizes for his molecular and clinical research and has authored and co-authored more that 200 publications.
Dr. Axel S. Merseburger is being referenced as one of the leading scientists involved in the research and development of Triptorelin. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Axel S. Merseburger is listed in [4] under the referenced citations.
Referenced Citations
- Breastcancer.org. Tamoxifen (SERMs) overview.
- F. Perrone et al., “Adjuvant zoledronic acid and letrozole plus ovarian function suppression in premenopausal breast cancer: HOBOE phase 3 randomised trial,” Eur. J. Cancer, Jun. 2019.
- J. E. Frampton, “Triptorelin: A Review of its Use as an Adjuvant Anticancer Therapy in Early Breast Cancer,” Drugs, vol. 77, no. 18, pp. 2037–2048, Dec. 2017.
- A. S. Merseburger and M. C. Hupe, “An Update on Triptorelin: Current Thinking on Androgen Deprivation Therapy for Prostate Cancer,” Adv. Ther., vol. 33, pp. 1072–1093, 2016.
- G. Marvaso, A. Viola, C. Fodor, and B. A. Jereczek-Fossa, “Radiotherapy Plus Total Androgen Block Versus Radiotherapy Plus LHRH Analog Monotherapy for Non-metastatic Prostate Cancer,” Anticancer Res., vol. 38, no. 5, pp. 3139–3143, 2018.
- K. Hachi et al., “[Study of the beneficial effects of triptorelin on lower urinary tract symptoms in Algeria in patients with non-localized prostate cancer],” Progres En Urol., vol. 28, no. 8–9, pp. 450–459, Jun. 2018.
- T. Gil, F. Aoun, P. Cabri, P. Maisonobe, and R. van Velthoven, “A prospective, observational grouped analysis to evaluate the effect of triptorelin on lower urinary tract symptoms in patients with advanced prostate cancer,” Ther. Adv. Urol., vol. 7, no. 3, pp. 116–124, Jun. 2015.
- M. Meli et al., “Triptorelin for Fertility Preservation in Adolescents Treated With Chemotherapy for Cancer,” J. Pediatr. Hematol. Oncol., vol. 40, no. 4, pp. 269–276, 2018.
- L. Del Mastro et al., “Effect of the gonadotropin-releasing hormone analogue triptorelin on the occurrence of chemotherapy-induced early menopause in premenopausal women with breast cancer: a randomized trial,” JAMA, vol. 306, no. 3, pp. 269–276, Jul. 2011.
- M. Xie, H. Yu, X. Zhang, W. Wang, and Y. Ren, “Elasticity of adenomyosis is increased after GnRHa therapy and is associated with spontaneous pregnancy in infertile patents,” J. Gynecol. Obstet. Hum. Reprod., May 2019.
- S. Lehrer, P. H. Rheinstein, and K. E. Rosenzweig, “No Relationship of Anti-Androgens to Alzheimer’s Disease or Cognitive Disorder in the MedWatch Database,” J. Alzheimers Dis. Rep., vol. 2, no. 1, pp. 123–127, Jun. 2018.
- U. Leone Roberti Maggiore et al., “Triptorelin for the treatment of endometriosis,” Expert Opin. Pharmacother., vol. 15, no. 8, pp. 1153–1179, Jun. 2014.
- H. Xue, M. Liu, W. Hao, and Y. Li, “Clinical evaluation of laparoscopic surgery combined with triptorelin acetate in patients with endometriosis and infertility,” Pak. J. Med. Sci., vol. 34, no. 5, pp. 1064–1069, Oct. 2018.
- B. Marchetti et al., “Luteinizing hormone-releasing hormone (LHRH) agonist restoration of age-associated decline of thymus weight, thymic LHRH receptors, and thymocyte proliferative capacity,” Endocrinology, vol. 125, no. 2, pp. 1037–1045, Aug. 1989.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.